Polycystic ovary syndrome (PCOS) is a condition characterized by the presence of hyperandrogenism (HA), oligo-anovulation, clinical and/or biochemical findings of and polycystic ovaries on ultrasound8. In our study, the PCOS patient group exhibited increased levels of oligomenorrhea-amenorrhea, hirsutism, the free androgen index, serum total testosterone, and dehydroepiandrosterone sulfate (DHEA-S) compared to the control group.
Mahde et al. reported biochemical HA in 43.5% and clinical HA in 60.9% of the cases9. Our findings revealed a rate of 76.9% for clinical HA and 66.7% for laboratory HA among our PCOS patient group. These results indicate that the majority of clinical HA cases are accompanied by laboratory HA, thereby supporting the clinical diagnosis.
Sachdeva et al. report more common insulin resistance among obese PCOS group10. In this study, correlation analysis of BMI as a marker of obesity and HOMA-IR as a marker of insulin resistance yielded statistical significance.
Elevated levels of calprotectin have been observed in patients with inflammatory pathologies. To ascertain the impact of insulin resistance on serum calprotectin levels, a study was conducted to examine the relationship between HOMA-IR, a marker of insulin resistance, and serum calprotectin values in a cohort of patients with polycystic ovary syndrome. Despite higher levels of serum calprotectin among high HOMA-IR patients, statistical significance was not achieved. Upon analyzing the correlation between HOMA-IR and serum calprotectin, it was found that although the calprotectin was higher in high HOMA-IR patients, the p value of 0.101 yielded no significant correlation.
Calprotectin is a significant marker of the inflammatory response, particularly as a protein released following neutrophil activation. Chronic low-grade inflammation is known to contribute to the pathophysiology of PCOS. Several studies indicate that patients with PCOS may exhibit elevated levels of proinflammatory cytokines and inflammatory markers, which are linked to insulin resistance, hyperandrogenemia, and metabolic disorders. It is believed that calprotectin may be influenced by these mechanisms, with insulin resistance stimulating its production through increased neutrophil activation, thereby intensifying the inflammatory response. Additionally, visceral adipose tissue is considered a key inflammatory source in PCOS, and the proinflammatory mediators released from this tissue may further contribute to the elevated levels of calprotectin. Some studies have demonstrated that elevated calprotectin levels may serve as a marker of catabolism in a multitude of inflammatory diseases11. In their investigation into the potential association between PCOS and calprotectin, Shouzhen Chen and colleagues observed that calprotectin levels were significantly higher in PCOS patients compared to controls12. Our findings also revealed significantly elevated serum calprotectin levels in the PCOS patient cohort. While these results highlight calprotectin as a potential marker in PCOS, further research is required to evaluate different phenotypes separately in conditions with multiple phenotypes, such as PCOS. The detection of patients with low or high serum calprotectin levels in the PCOS group may take on a new meaning with further investigation of subgroups. Therefore, calprotectin can be used more efficiently.
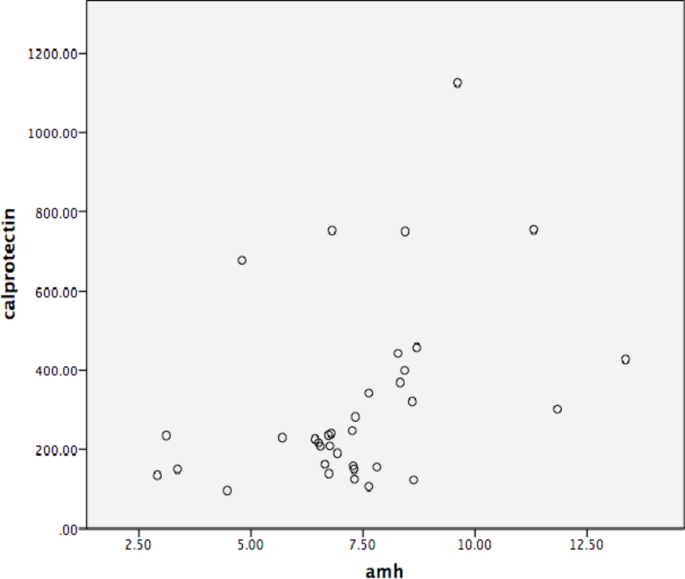
Laven et al.13 observed a significant positive correlation between serum AMH levels and LH, testosterone, androstenedione, free androgen index, and number of follicles. Our study revealed a significant positive correlation between serum calprotectin levels and serum AMH levels, as demonstrated by correlation analysis. The correlation of calprotectin levels with AMH levels, an established marker that has been shown to provide significant clinical information in PCOS, is a valuable finding. This condition necessitated a comparison between the sensitivity and specificity of these two markers in PCOS. In their study evaluating AMH in PCOS, Le et al.14 found 78.50% sensitivity and 75.83% specificity. In our study, the AMH cut-off value for PCOS was determined to be 4.34 ng/mL, resulting in a sensitivity of 90.40% and a specificity of 69.20%. Similarly, a serum calprotectin cut-off value of 204.54 pg/mL yielded a sensitivity of 66.70% and a specificity of 54.70%. These results indicate that neither marker is sufficient for PCOS diagnosis on its own, and that both can be used in conjunction with clinical and laboratory evidence for diagnosis. In contrast, Chen et al.12 report a sensitivity of 75.6% and specificity of 85.2% in identifying PCOS for serum calprotectin. While they found a higher sensitivity, this is still below the levels for AMH. Therefore, it is not possible to conclude with certainty that AMH outperforms calprotectin in the diagnosis of PCOS. The extensive prior use of AMH in studies and clinical settings allows for the collection of larger data sets. A meta-analysis involving future calprotectin studies may provide an objective evaluation.
Although the study provides strong data with its prospective design, it has limitations that may arise from a single centre and small sample size. Due to the multivariate clinical nature of PCOS, a large subgroup analysis can be performed in the evaluation of the parameters and the results can be more meaningfully examined for diagnostic performance. In addition, as there were not enough studies evaluating calprotectin performance in PCOS during the study period, the analysis compared the results with other previously investigated parameters.
link