GBD Chronic Kidney Disease Collaboration. Global, regional, and national burden of chronic kidney disease, 1990-2017: a systematic analysis for the Global Burden of Disease Study 2017. Lancet 395, 709–733 (2020).
Google Scholar
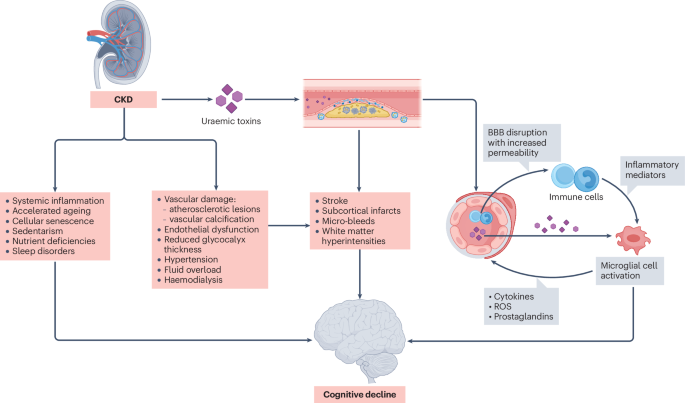
Viggiano, D. et al. Mechanisms of cognitive dysfunction in CKD. Nat. Rev. Nephrol. 16, 452–469 (2020).
Google Scholar
de Boer, A. et al. Consensus-based technical recommendations for clinical translation of renal phase contrast MRI. J. Magn. Reson. Imaging 55, 323–335 (2022).
Google Scholar
Wikoff, W. R., Nagle, M. A., Kouznetsova, V. L., Tsigelny, I. F. & Nigam, S. K. Untargeted metabolomics identifies enterobiome metabolites and putative uremic toxins as substrates of organic anion transporter 1 (Oat1). J. Proteome Res. 10, 2842–2851 (2011).
Google Scholar
Rosner, M. H. et al. Classification of uremic toxins and their role in kidney failure. Clin. J. Am. Soc. Nephrol. 16, 1918–1928 (2021).
Google Scholar
Perna, A. F. et al. Increased plasma protein homocysteinylation in hemodialysis patients. Kidney Int. 69, 869–876 (2006).
Google Scholar
Karbowska, M. et al. Neurobehavioral effects of uremic toxin-indoxyl sulfate in the rat model. Sci. Rep. 10, 9483 (2020).
Google Scholar
Wulczyn, K. E. et al. Metabolites associated with uremic symptoms in patients with CKD: findings from the chronic renal insufficiency cohort (CRIC) study. Am. J. Kidney Dis. 84, 49–61.e1 (2024).
Google Scholar
Al Awadhi, S. et al. A metabolomics approach to identify metabolites associated with mortality in patients receiving maintenance hemodialysis. Kidney Int. Rep. 9, 2718–2726 (2024).
Google Scholar
Xu, S. et al. Cognitive impairment in chronic kidney disease is associated with glymphatic system dysfunction. Kidney Dis. 9, 384–397 (2023).
Google Scholar
Kelly, D. M. et al. Impaired kidney function, cerebral small vessel disease and cognitive disorders: the Framingham Heart Study. Nephrol. Dial. Transpl. 39, 1911–1922 (2024).
Google Scholar
Pépin, M. et al. Cognitive disorders in patients with chronic kidney disease: approaches to prevention and treatment. Eur. J. Neurol. 30, 2899–2911 (2023).
Google Scholar
De Deyn, P. P., Vanholder, R., Eloot, S. & Glorieux, G. Guanidino compounds as uremic (neuro)toxins. Semin. Dial. 22, 340–345 (2009).
Google Scholar
Meera, P., Uusi-Oukari, M., Wallner, M. & Lipshutz, G. S. Guanidinoacetate (GAA) is a potent GABAA receptor GABA mimetic: implications for neurological disease pathology. J. Neurochem. 165, 445–454 (2023).
Google Scholar
Greenfield, L. J. Jr. Molecular mechanisms of antiseizure drug activity at GABAA receptors. Seizure 22, 589–600 (2013).
Google Scholar
Yeh, Y. C. et al. Indoxyl sulfate, not p-cresyl sulfate, is associated with cognitive impairment in early-stage chronic kidney disease. Neurotoxicology 53, 148–152 (2016).
Google Scholar
Jeong, S. H. et al. Indoxyl sulfate induces apoptotic cell death by inhibiting glycolysis in human astrocytes. Kidney Res. Clin. Pract. 43, 774–784 (2023).
Google Scholar
Sato, E. et al. Impact of the oral adsorbent AST-120 on organ-specific accumulation of uremic toxins: LC-MS/MS and MS imaging techniques. Toxins (2017).
Faucher, Q., van der Made, T. K., De Lange, E. & Masereeuw, R. Blood–brain barrier perturbations by uremic toxins: key contributors in chronic kidney disease-induced neurological disorders? Eur. J. Pharm. Sci. 187, 106462 (2023).
Google Scholar
Chai, G. S. et al. H3K4 trimethylation mediate hyperhomocysteinemia induced neurodegeneration via suppressing histone acetylation by ANP32A. Mol. Neurobiol. 61, 6788–6804 (2024).
Google Scholar
Ingrosso, D. & Perna, A. F. DNA methylation dysfunction in chronic kidney disease. Genes (2020).
Perna, A. F. et al. Plasma protein aspartyl damage is increased in hemodialysis patients: studies on causes and consequences. J. Am. Soc. Nephrol. 15, 2747–2754 (2004).
Google Scholar
Tang, W. H. et al. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N. Engl. J. Med. 368, 1575–1584 (2013).
Google Scholar
Li, D. et al. Trimethylamine-N-oxide promotes brain aging and cognitive impairment in mice. Aging Cell 17, e12768 (2018).
Google Scholar
Morrens, M. The role of the kynurenine pathway in cognitive function in brain disorders: insights and challenges. Brain Behav. Immun. 127, 110–111 (2025).
Google Scholar
Deora, G. S. et al. Multifunctional analogs of kynurenic acid for the treatment of Alzheimer’s disease: synthesis, pharmacology, and molecular modeling studies. ACS Chem. Neurosci. 8, 2667–2675 (2017).
Google Scholar
Szabo, N., Kincses, Z. T., Toldi, J. & Vecsei, L. Altered tryptophan metabolism in Parkinson’s disease: a possible novel therapeutic approach. J. Neurol. Sci. 310, 256–260 (2011).
Google Scholar
Zadori, D. et al. Neuroprotective effects of a novel kynurenic acid analogue in a transgenic mouse model of Huntington’s disease. J. Neural Transm. 118, 865–875 (2011).
Google Scholar
Fukui, S., Schwarcz, R., Rapoport, S. I., Takada, Y. & Smith, Q. R. Blood-brain barrier transport of kynurenines: implications for brain synthesis and metabolism. J. Neurochem. 56, 2007–2017 (1991).
Google Scholar
Vezzani, A. et al. Studies on the potential neurotoxic and convulsant effects of increased blood levels of quinolinic acid in rats with altered blood–brain barrier permeability. Exp. Neurol. 106, 90–98 (1989).
Google Scholar
Rahman, A. et al. The excitotoxin quinolinic acid induces tau phosphorylation in human neurons. PLoS ONE 4, e6344 (2009).
Google Scholar
Köhler, C. et al. Quinolinic acid metabolism in the rat brain. Immunohistochemical identification of 3-hydroxyanthranilic acid oxygenase and quinolinic acid phosphoribosyltransferase in the hippocampal region. J. Neurosci. 8, 975–987 (1988).
Google Scholar
Jankowski, J., Floege, J., Fliser, D., Böhm, M. & Marx, N. Cardiovascular disease in chronic kidney disease: pathophysiological insights and therapeutic options. Circulation 143, 1157–1172 (2021).
Google Scholar
Kelly, D. M. et al. Chronic kidney disease and cerebrovascular disease: consensus and guidance from a KDIGO controversies conference. Stroke 52, e328–e346 (2021).
Google Scholar
Liu, Y. et al. Association between low estimated glomerular filtration rate and risk of cerebral small-vessel diseases: a meta-analysis. J. Stroke Cerebrovasc. Dis. 25, 710–716 (2016).
Google Scholar
Ter Telgte, A. et al. Cerebral small vessel disease: from a focal to a global perspective. Nat. Rev. Neurol. 14, 387–398 (2018).
Google Scholar
Sedaghat, S. et al. The AGES-Reykjavik Study suggests that change in kidney measures is associated with subclinical brain pathology in older community-dwelling persons. Kidney Int. 94, 608–615 (2018).
Google Scholar
Zheng, G. et al. Elevated global cerebral blood flow, oxygen extraction fraction and unchanged metabolic rate of oxygen in young adults with end-stage renal disease: an MRI study. Eur. Radiol. 26, 1732–1741 (2016).
Google Scholar
Tamura, M. K. et al. Chronic kidney disease, cerebral blood flow, and white matter volume in hypertensive adults. Neurology 86, 1208–1216 (2016).
Google Scholar
Wang, H. et al. Dysfunctional coupling of cerebral blood flow and susceptibility value in the bilateral hippocampus is associated with cognitive decline in nondialysis patients with CKD. J. Am. Soc. Nephrol. 34, 1574–1588 (2023).
Google Scholar
Song, L. et al. Combination of rs-fMRI, QSM, and ASL reveals the cerebral neurovascular coupling dysfunction is associated with cognitive decline in patients with chronic kidney disease. CNS Neurosci. Ther. 30, e70151 (2024).
Google Scholar
MacEwen, C., Sutherland, S., Daly, J., Pugh, C. & Tarassenko, L. Relationship between hypotension and cerebral ischemia during hemodialysis. J. Am. Soc. Nephrol. 28, 2511–2520 (2017).
Google Scholar
Inserra, F., Forcada, P., Castellaro, A. & Castellaro, C. Chronic kidney disease and arterial stiffness: a two-way path. Front. Med. 8, 765924 (2021).
Google Scholar
Dusing, P. et al. Vascular pathologies in chronic kidney disease: pathophysiological mechanisms and novel therapeutic approaches. J. Mol. Med. 99, 335–348 (2021).
Google Scholar
Roumeliotis, S., Mallamaci, F. & Zoccali, C. Endothelial dysfunction in chronic kidney disease, from biology to clinical outcomes: a 2020 update. J. Clin. Med. (2020).
Stoddart, P., Satchell, S. C. & Ramnath, R. Cerebral microvascular endothelial glycocalyx damage, its implications on the blood–brain barrier and a possible contributor to cognitive impairment. Brain Res. 1780, 147804 (2022).
Google Scholar
Kruger, A. et al. Laser Doppler flowmetry detection of endothelial dysfunction in end-stage renal disease patients: correlation with cardiovascular risk. Kidney Int. 70, 157–164 (2006).
Google Scholar
Dane, M. J. et al. Association of kidney function with changes in the endothelial surface layer. Clin. J. Am. Soc. Nephrol. 9, 698–704 (2014).
Google Scholar
Vlahu, C. A. et al. Damage of the endothelial glycocalyx in dialysis patients. J. Am. Soc. Nephrol. 23, 1900–1908 (2012).
Google Scholar
Oberleithner, H. et al. Plasma sodium stiffens vascular endothelium and reduces nitric oxide release. Proc. Natl Acad. Sci. USA 104, 16281–16286 (2007).
Google Scholar
Bobot, M. et al. Uremic toxic blood–brain barrier disruption mediated by AhR activation leads to cognitive impairment during experimental renal dysfunction. J. Am. Soc. Nephrol. 31, 1509–1521 (2020).
Google Scholar
Gupta, A. et al. Blood-brain barrier permeability in ESKD – a proof-of-concept study. J. Am. Soc. Nephrol. 34, 1508–1511 (2023).
Google Scholar
Bobot, M. et al. Increased blood–brain barrier permeability and cognitive impairment in patients with ESKD. Kidney Int. Rep. 9, 2988–2995 (2024).
Google Scholar
Matsuki, H. et al. Chronic kidney disease causes blood–brain barrier breakdown via urea-activated matrix metalloproteinase-2 and insolubility of tau protein. Aging 15, 10972–10995 (2023).
Google Scholar
Hernandez, L. et al. Blood–brain barrier and gut barrier dysfunction in chronic kidney disease with a focus on circulating biomarkers and tight junction proteins. Sci. Rep. 12, 4414 (2022).
Google Scholar
Sweeney, M. D., Sagare, A. P. & Zlokovic, B. V. Blood–brain barrier breakdown in Alzheimer disease and other neurodegenerative disorders. Nat. Rev. Neurol. 14, 133–150 (2018).
Google Scholar
Crowe, K., Quinn, T. J., Mark, P. B. & Findlay, M. D. “Is it removed during dialysis?” – Cognitive dysfunction in advanced kidney failure – a review article. Front. Neurol. 12, 787370 (2021).
Google Scholar
Kurella Tamura, M. et al. Loss of executive function after dialysis initiation in adults with chronic kidney disease. Kidney Int. 91, 948–953 (2017).
Google Scholar
Iyasere, O., Okai, D. & Brown, E. Cognitive function and advanced kidney disease: longitudinal trends and impact on decision-making. Clin. Kidney J. 10, 89–94 (2017).
Google Scholar
Eldehni, M. T., Odudu, A. & McIntyre, C. W. Randomized clinical trial of dialysate cooling and effects on brain white matter. J. Am. Soc. Nephrol. 26, 957–965 (2015).
Google Scholar
Murray, A. M., Seliger, S., Lakshminarayan, K., Herzog, C. A. & Solid, C. A. Incidence of stroke before and after dialysis initiation in older patients. J. Am. Soc. Nephrol. 24, 1166–1173 (2013).
Google Scholar
Neumann, D., Mau, W., Wienke, A. & Girndt, M. Peritoneal dialysis is associated with better cognitive function than hemodialysis over a one-year course. Kidney Int. 93, 430–438 (2018).
Google Scholar
Polinder-Bos, H. A. et al. Hemodialysis induces an acute decline in cerebral blood flow in elderly patients. J. Am. Soc. Nephrol. 29, 1317–1325 (2018).
Google Scholar
Anazodo, U. C. et al. Hemodialysis-related acute brain injury demonstrated by application of intradialytic magnetic resonance imaging and spectroscopy. J. Am. Soc. Nephrol. 34, 1090–1104 (2023).
Google Scholar
Richerson, W. T., Schmit, B. D. & Wolfgram, D. F. The relationship between cerebrovascular reactivity and cerebral oxygenation during hemodialysis. J. Am. Soc. Nephrol. 33, 1602–1612 (2022).
Google Scholar
Lepping, R. J. et al. Normalization of cerebral blood flow, neurochemicals, and white matter integrity after kidney transplantation. J. Am. Soc. Nephrol. 32, 177–187 (2021).
Google Scholar
Berger, I. et al. Cognition in chronic kidney disease: a systematic review and meta-analysis. BMC Med. 14, 206 (2016).
Google Scholar
Harada, C. N., Natelson Love, M. C. & Triebel, K. L. Normal cognitive aging. Clin. Geriatr. Med. 29, 737–752 (2013).
Google Scholar
Kooman, J. P., Kotanko, P., Schols, A. M., Shiels, P. G. & Stenvinkel, P. Chronic kidney disease and premature ageing. Nat. Rev. Nephrol. 10, 732–742 (2014).
Google Scholar
Dai, L., Schurgers, L. J., Shiels, P. G. & Stenvinkel, P. Early vascular ageing in chronic kidney disease: impact of inflammation, vitamin K, senescence and genomic damage. Nephrol. Dial. Transpl. 35, ii31–ii37 (2020).
Google Scholar
McHugh, D. & Gil, J. Senescence and aging: causes, consequences, and therapeutic avenues. J. Cell Biol. 217, 65–77 (2018).
Google Scholar
Di Micco, R., Krizhanovsky, V., Baker, D. & d’Adda di Fagagna, F. Cellular senescence in ageing: from mechanisms to therapeutic opportunities. Nat. Rev. Mol. Cell Biol. 22, 75–95 (2021).
Google Scholar
Sikora, E. et al. Cellular senescence in brain aging. Front. Aging Neurosci. 13, 646924 (2021).
Google Scholar
Hu, Y. et al. Replicative senescence dictates the emergence of disease-associated microglia and contributes to Aβ pathology. Cell Rep. 35, 109228 (2021).
Google Scholar
Lau, V., Ramer, L. & Tremblay, M. E. An aging, pathology burden, and glial senescence build-up hypothesis for late onset Alzheimer’s disease. Nat. Commun. 14, 1670 (2023).
Google Scholar
Angelova, D. M. & Brown, D. R. Altered processing of β-amyloid in SH-SY5Y cells induced by model senescent microglia. ACS Chem. Neurosci. 9, 3137–3152 (2018).
Google Scholar
McAvoy, K. M. et al. Modulating neuronal competition dynamics in the dentate gyrus to rejuvenate aging memory circuits. Neuron 91, 1356–1373 (2016).
Google Scholar
Ogrodnik, M. et al. Whole-body senescent cell clearance alleviates age-related brain inflammation and cognitive impairment in mice. Aging Cell 20, e13296 (2021).
Google Scholar
Kyriakidis, N. C., Cobo, G., Dai, L., Lindholm, B. & Stenvinkel, P. Role of uremic toxins in early vascular ageing and calcification. Toxins (2021).
Assem, M. et al. The impact of uremic toxins on cerebrovascular and cognitive disorders. Toxins (2018).
Jaber, B. L. et al. Apoptosis of leukocytes: basic concepts and implications in uremia. Kidney Int. Suppl. 78, S197–S205 (2001).
Google Scholar
Laville, S. M. et al. Urea levels and cardiovascular disease in patients with chronic kidney disease. Nephrol. Dial. Transpl. 38, 184–192 (2022).
Google Scholar
Sun, C. Y., Chang, S. C. & Wu, M. S. Suppression of Klotho expression by protein-bound uremic toxins is associated with increased DNA methyltransferase expression and DNA hypermethylation. Kidney Int. 81, 640–650 (2012).
Google Scholar
Neytchev, O. et al. Epigenetic clocks indicate that kidney transplantation and not dialysis mitigate the effects of renal ageing. J. Intern. Med. 295, 79–90 (2024).
Google Scholar
Shaker, M. R., Aguado, J., Chaggar, H. K. & Wolvetang, E. J. Klotho inhibits neuronal senescence in human brain organoids. NPJ Aging Mech. Dis. 7, 18 (2021).
Google Scholar
Chen, W. T. et al. Impaired leukocytes autophagy in chronic kidney disease patients. Cardiorenal Med. 3, 254–264 (2013).
Google Scholar
Lin, T. A., Wu, V. C. & Wang, C. Y. Autophagy in chronic kidney diseases. Cells (2019).
Dai, X. Y. et al. Phosphate-induced autophagy counteracts vascular calcification by reducing matrix vesicle release. Kidney Int. 83, 1042–1051 (2013).
Google Scholar
Tsirpanlis, G. et al. Telomerase activity is decreased in peripheral blood mononuclear cells of hemodialysis patients. Am. J. Nephrol. 26, 91–96 (2006).
Google Scholar
Miyamoto, M. I., Djabali, K. & Gordon, L. B. Atherosclerosis in ancient humans, accelerated aging syndromes and normal aging: is lamin A protein a common link? Glob. Heart 9, 211–218 (2014).
Google Scholar
Liu, Y., Drozdov, I., Shroff, R., Beltran, L. E. & Shanahan, C. M. Prelamin A accelerates vascular calcification via activation of the DNA damage response and senescence-associated secretory phenotype in vascular smooth muscle cells. Circ. Res. 112, e99–e109 (2013).
Google Scholar
Gonzales, M. M. et al. Senolytic therapy to modulate the progression of Alzheimer’s disease (SToMP-AD): a pilot clinical trial. J. Prev. Alzheimer’s Dis. 9, 22–29 (2022).
Google Scholar
Zhang, F., Yin, X., Huang, L. & Zhang, H. The “adult inactivity triad” in patients with chronic kidney disease: a review. Front. Med. 10, 1160450 (2023).
Google Scholar
Raichlen, D. A. et al. Sedentary behavior and incident dementia among older adults. JAMA 330, 934–940 (2023).
Google Scholar
Zoccali, C. et al. Cardiovascular complications in chronic kidney disease: a review from the European Renal and Cardiovascular Medicine Working Group of the European Renal Association. Cardiovasc. Res. 119, 2017–2032 (2023).
Google Scholar
Jia, R. et al. Cardiovascular disease risk models and dementia or cognitive decline: a systematic review. Front. Aging Neurosci. (2023).
Manfredini, F. et al. Exercise in patients on dialysis: a multicenter, randomized clinical trial. J. Am. Soc. Nephrol. 28, 1259–1268 (2017).
Google Scholar
Mandolesi, L. et al. Environmental factors promoting neural plasticity: insights from animal and human studies. Neural Plast. 2017, 7219461 (2017).
Google Scholar
Loprinzi, P. D., Moore, D. & Loenneke, J. P. Does aerobic and resistance exercise influence episodic memory through unique mechanisms? Brain Sci. (2020).
Sartori, A. C., Vance, D. E., Slater, L. Z. & Crowe, M. The impact of inflammation on cognitive function in older adults: implications for healthcare practice and research. J. Neurosci. Nurs. 44, 206–217 (2012).
Google Scholar
Bishop, N. C. et al. Exercise and chronic kidney disease: potential mechanisms underlying the physiological benefits. Nat. Rev. Nephrol. 19, 244–256 (2023).
Google Scholar
Rotondi, S. et al. Association between cognitive impairment and malnutrition in hemodialysis patients: two sides of the same coin. Nutrients (2023).
Samson, M. E. et al. Vitamin B-12 malabsorption and renal function are critical considerations in studies of folate and vitamin B-12 interactions in cognitive performance: NHANES 2011-2014. Am. J. Clin. Nutr. 116, 74–85 (2022).
Google Scholar
Hossain, M. F. et al. Prevalence of electrolyte imbalance in patients with acute stroke: a systematic review. Cureus 15, e43149 (2023).
Google Scholar
Zoccali, C. & Mallamaci, F. Mapping progress in reducing cardiovascular risk with kidney disease: managing volume overload. Clin. J. Am. Soc. Nephrol. 13, 1432–1434 (2018).
Google Scholar
Otobe, Y., Hiraki, K., Izawa, K. P., Sakurada, T. & Shibagaki, Y. Relationship between serum inorganic phosphorus levels and cognitive decline over 2 years in older adults with pre-dialysis chronic kidney disease. Clin. Exp. Nephrol. 24, 286–287 (2020).
Google Scholar
Deak, M. C. & Stickgold, R. Sleep and cognition. Wiley Interdiscip. Rev. Cogn. Sci. 1, 491–500 (2010).
Google Scholar
Wong, R. & Lovier, M. A. Sleep disturbances and dementia risk in older adults: findings from 10 years of National U.S. prospective data. Am. J. Prev. Med. 64, 781–787 (2023).
Google Scholar
Long, J. J. et al. Sleep disorders and dementia risk in older patients with kidney failure: a retrospective cohort study. Clin. J. Am. Soc. Nephrol. 19, 1301–1309 (2024).
Google Scholar
Maung, S. C., El Sara, A., Chapman, C., Cohen, D. & Cukor, D. Sleep disorders and chronic kidney disease. World J. Nephrol. 5, 224–232 (2016).
Google Scholar
Lyons, O. D. Sleep disorders in chronic kidney disease. Nat. Rev. Nephrol. 20, 690–700 (2024).
Google Scholar
Merchant, R. A. & Vathsala, A. Healthy aging and chronic kidney disease. Kidney Res. Clin. Pract. 41, 644–656 (2022).
Google Scholar
Tan, L. H. et al. Insomnia and poor sleep in CKD: a systematic review and meta-analysis. Kidney Med. 4, 100458 (2022).
Google Scholar
Pierratos, A. & Hanly, P. J. Sleep disorders over the full range of chronic kidney disease. Blood Purif. 31, 146–150 (2011).
Google Scholar
Pai, M. F. et al. Sleep disturbance in chronic hemodialysis patients: the impact of depression and anemia. Ren. Fail. 29, 673–677 (2007).
Google Scholar
Parker, K. P. Sleep and dialysis: a research-based review of the literature. ANNA J. 24, 626–639 (1997).
Google Scholar
Madero, M., Gul, A. & Sarnak, M. J. Cognitive function in chronic kidney disease. Semin. Dial. 21, 29–37 (2008).
Google Scholar
You, J. et al. Clinical trajectories preceding incident dementia up to 15 years before diagnosis: a large prospective cohort study. Mol. Psychiatry 29, 3097–3105 (2024).
Google Scholar
Jessen, N. A., Munk, A. S., Lundgaard, I. & Nedergaard, M. The glymphatic system: a beginner’s guide. Neurochem. Res. 40, 2583–2599 (2015).
Google Scholar
Tarasoff-Conway, J. M. et al. Clearance systems in the brain – implications for Alzheimer disease. Nat. Rev. Neurol. 11, 457–470 (2015).
Google Scholar
Louveau, A. et al. Structural and functional features of central nervous system lymphatic vessels. Nature 523, 337–341 (2015).
Google Scholar
Plog, B. A. et al. Biomarkers of traumatic injury are transported from brain to blood via the glymphatic system. J. Neurosci. 35, 518–526 (2015).
Google Scholar
Rasmussen, M. K., Mestre, H. & Nedergaard, M. Fluid transport in the brain. Physiol. Rev. 102, 1025–1151 (2022).
Google Scholar
Xie, L. et al. Sleep drives metabolite clearance from the adult brain. Science 342, 373–377 (2013).
Google Scholar
Eide, P. K., Vinje, V., Pripp, A. H., Mardal, K. A. & Ringstad, G. Sleep deprivation impairs molecular clearance from the human brain. Brain : J. Neurol. 144, 863–874 (2021).
Google Scholar
Helakari, H. et al. Human NREM sleep promotes brain-wide vasomotor and respiratory pulsations. J. Neurosci. 42, 2503–2515 (2022).
Google Scholar
Nedergaard, M. & Goldman, S. A. Glymphatic failure as a final common pathway to dementia. Science 370, 50–56 (2020).
Google Scholar
Rose, K. M. & Lorenz, R. Sleep disturbances in dementia. J. Gerontol. Nurs. 36, 9–14 (2010).
Google Scholar
Taoka, T. et al. Diffusion tensor image analysis along the perivascular space (DTI-ALPS): revisiting the meaning and significance of the method. Magn. Reson. Med. Sci. 23, 268–290 (2024).
Google Scholar
Heo, C. M. et al. Glymphatic dysfunction in patients with end-stage renal disease. Front. Neurol. 12, 809438 (2021).
Google Scholar
Lee, Y. J. et al. Changes in the glymphatic system before and after dialysis initiation in patients with end-stage kidney disease. Ren. Fail. 45, 2265665 (2023).
Google Scholar
Heo, C. M. et al. Glymphatic system dysfunction in patients with early chronic kidney disease. Front. Neurol. 13, 976089 (2022).
Google Scholar
Yazıcı, R. & Güney, İ. Prevalence and related factors of poor sleep quality in patients with pre-dialysis chronic kidney disease. Int. J. Artif. Organs 45, 905–910 (2022).
Google Scholar
Hasanzamani, B., Pourranjbar, E. & Rezaei Ardani, A. Comparing sleep quality in patients before and after kidney transplantation. Iran. J. Kidney Dis. 14, 139–144 (2020).
Google Scholar
Zhang, C. Y., He, F. F., Su, H., Zhang, C. & Meng, X. F. Association between chronic kidney disease and Alzheimer’s disease: an update. Metab. Brain Dis. 35, 883–894 (2020).
Google Scholar
Wardlaw, J. M. et al. Perivascular spaces in the brain: anatomy, physiology and pathology. Nat. Rev. Neurol. 16, 137–153 (2020).
Google Scholar
Kurella, M., Chertow, G. M., Luan, J. & Yaffe, K. Cognitive impairment in chronic kidney disease. J. Am. Geriatr. Soc. 52, 1863–1869 (2004).
Google Scholar
Qiu, Y. et al. Structural and functional brain alterations in end stage renal disease patients on routine hemodialysis: a voxel-based morphometry and resting state functional connectivity study. PLoS ONE 9, e98346 (2014).
Google Scholar
Nowak, K. L. & Chonchol, M. Does inflammation affect outcomes in dialysis patients? Semin. Dial. 31, 388–397 (2018).
Google Scholar
Sarnak, M. J. & Jaber, B. L. Mortality caused by sepsis in patients with end-stage renal disease compared with the general population. Kidney Int. 58, 1758–1764 (2000).
Google Scholar
Verkade, M. A. et al. Peripheral blood dendritic cells and GM-CSF as an adjuvant for hepatitis B vaccination in hemodialysis patients. Kidney Int. 66, 614–621 (2004).
Google Scholar
Stewart, J. H. et al. The pattern of excess cancer in dialysis and transplantation. Nephrol. Dial. Transpl. 24, 3225–3231 (2009).
Google Scholar
Betjes, M. G. Immune cell dysfunction and inflammation in end-stage renal disease. Nat. Rev. Nephrol. 9, 255–265 (2013).
Google Scholar
Qi, H., Yang, S. & Zhang, L. Neutrophil extracellular traps and endothelial dysfunction in atherosclerosis and thrombosis. Front. Immunol. 8, 928 (2017).
Google Scholar
Mutua, V. & Gershwin, L. J. A review of neutrophil extracellular traps (NETs) in disease: potential anti-NETs therapeutics. Clin. Rev. Allergy Immunol. 61, 194–211 (2021).
Google Scholar
Lee, H. W. et al. Uremic serum damages endothelium by provoking excessive neutrophil extracellular trap formation. Sci. Rep. 11, 21439 (2021).
Google Scholar
Nongnuch, A., Panorchan, K. & Davenport, A. Brain-kidney crosstalk. Crit. Care 18, 225 (2014).
Google Scholar
De Sousa, R. A. L. & Cassilhas, R. C. Microglia role as the regulator of cognitive function. Rev. Assoc. Med. Bras. 69, e20230412 (2023).
Google Scholar
Adesso, S. et al. Indoxyl sulfate affects glial function increasing oxidative stress and neuroinflammation in chronic kidney disease: interaction between astrocytes and microglia. Front. Pharmacol. 8, 370 (2017).
Google Scholar
Kurella Tamura, M. et al. Inflammatory markers and risk for cognitive decline in chronic kidney disease: the CRIC study. Kidney Int. Rep. 2, 192–200 (2017).
Google Scholar
Montinaro, V. et al. Emotional symptoms, quality of life and cytokine profile in hemodialysis patients. Clin. Nephrol. 73, 36–43 (2010).
Google Scholar
Zhu, B. et al. Abnormalities in gut microbiota and serum metabolites in hemodialysis patients with mild cognitive decline: a single-center observational study. Psychopharmacology 237, 2739–2752 (2020).
Google Scholar
Wang, J. et al. Correlation between gut microbiome and cognitive impairment in patients undergoing peritoneal dialysis. BMC Nephrol. 24, 360 (2023).
Google Scholar
Wang, Y. F. et al. The gut microbiota–inflammation–brain axis in end-stage renal disease: perspectives from default mode network. Theranostics 9, 8171–8181 (2019).
Google Scholar
Tracey, K. J. The inflammatory reflex. Nature 420, 853–859 (2002).
Google Scholar
Okusa, M. D., Rosin, D. L. & Tracey, K. J. Targeting neural reflex circuits in immunity to treat kidney disease. Nat. Rev. Nephrol. 13, 669–680 (2017).
Google Scholar
Sara, S. J. The locus coeruleus and noradrenergic modulation of cognition. Nat. Rev. Neurosci. 10, 211–223 (2009).
Google Scholar
Feinstein, D. L. et al. Noradrenergic regulation of inflammatory gene expression in brain. Neurochem. Int. 41, 357–365 (2002).
Google Scholar
Vonck, K. et al. Vagus nerve stimulation…25 years later! What do we know about the effects on cognition? Neurosci. Biobehav. Rev. 45, 63–71 (2014).
Google Scholar
Boon, P., Moors, I., De Herdt, V. & Vonck, K. Vagus nerve stimulation and cognition. Seizure 15, 259–263 (2006).
Google Scholar
Hu, M. C. et al. Klotho deficiency causes vascular calcification in chronic kidney disease. J. Am. Soc. Nephrol. 22, 124–136 (2011).
Google Scholar
Fernandez-Fernandez, B. et al. Albumin downregulates Klotho in tubular cells. Nephrol. Dial. Transpl. 33, 1712–1722 (2018).
Google Scholar
Nagai, T. et al. Cognition impairment in the genetic model of aging klotho gene mutant mice: a role of oxidative stress. FASEB J. 17, 50–52 (2003).
Google Scholar
Kuro-o, M. et al. Mutation of the mouse klotho gene leads to a syndrome resembling ageing. Nature 390, 45–51 (1997).
Google Scholar
Lindberg, K. et al. The kidney is the principal organ mediating klotho effects. J. Am. Soc. Nephrol. 25, 2169–2175 (2014).
Google Scholar
Ohnishi, M. & Razzaque, M. S. Dietary and genetic evidence for phosphate toxicity accelerating mammalian aging. FASEB J. 24, 3562–3571 (2010).
Google Scholar
Dronsfield, A. & Ellis, P. Phosphorus – food for thought. Educ. Chem. edu.rsc.org/feature/phosphorus-food-for-thought/2020208.article (2009).
Mora-Fernandez, C. et al. Sodium-glucose co-transporter-2 inhibitors increase Klotho in patients with diabetic kidney disease: a clinical and experimental study. Biomed. Pharmacother. 154, 113677 (2022).
Google Scholar
Kanbay, M. et al. Role of Klotho in the development of essential hypertension. Hypertension 77, 740–750 (2021).
Google Scholar
Favero, C., Pintor-Chocano, A., Sanz, A., Ortiz, A. & Sanchez-Nino, M. D. Butyrate promotes kidney resilience through a coordinated kidney protective response in tubular cells. Biochem. Pharmacol. 224, 116203 (2024).
Google Scholar
Valino-Rivas, L. et al. Growth differentiation factor-15 preserves Klotho expression in acute kidney injury and kidney fibrosis. Kidney Int. 101, 1200–1215 (2022).
Google Scholar
Cuarental, L. et al. The transcription factor Fosl1 preserves Klotho expression and protects from acute kidney injury. Kidney Int. 103, 686–701 (2023).
Google Scholar
Navarro-Gonzalez, J. F. et al. Effects of pentoxifylline on soluble klotho concentrations and renal tubular cell expression in diabetic kidney disease. Diabetes Care 41, 1817–1820 (2018).
Google Scholar
Kanbay, M. et al. Klotho: a potential therapeutic target in aging and neurodegeneration beyond chronic kidney disease – a comprehensive review from the ERA CKD-MBD working group. Clin. Kidney J. 17, sfad276 (2024).
Google Scholar
Zhu, L. et al. Klotho controls the brain–immune system interface in the choroid plexus. Proc. Natl Acad. Sci. USA 115, E11388–E11396 (2018).
Google Scholar
Dubnov, S. et al. Knockout of the longevity gene Klotho perturbs aging and Alzheimer’s disease-linked brain microRNAs and tRNA fragments. Commun. Biol. 7, 720 (2024).
Google Scholar
Leiter, O. et al. Platelet-derived exerkine CXCL4/platelet factor 4 rejuvenates hippocampal neurogenesis and restores cognitive function in aged mice. Nat. Commun. 14, 4375 (2023).
Google Scholar
Schroer, A. B. et al. Platelet factors attenuate inflammation and rescue cognition in ageing. Nature 620, 1071–1079 (2023).
Google Scholar
Park, C. et al. Platelet factors are induced by longevity factor klotho and enhance cognition in young and aging mice. Nat. Aging 3, 1067–1078 (2023).
Google Scholar
Duce, J. A. et al. Gene profile analysis implicates Klotho as an important contributor to aging changes in brain white matter of the rhesus monkey. Glia 56, 106–117 (2008).
Google Scholar
Castner, S. A. et al. Longevity factor klotho enhances cognition in aged nonhuman primates. Nat. Aging 3, 931–937 (2023).
Google Scholar
Kuriyama, N. et al. Association between α-klotho and deep white matter lesions in the brain: a pilot case control study using brain MRI. J. Alzheimers Dis. 61, 145–155 (2018).
Google Scholar
Wu, Y. et al. Relationship of Klotho with cognition and dementia: results from the NHANES 2011-2014 and Mendelian randomization study. Transl. Psychiatry 13, 337 (2023).
Google Scholar
Blizzard LeBlanc, D. R. et al. Exercise-induced irisin release as a determinant of the metabolic response to exercise training in obese youth: the EXIT trial. Physiol. Rep. (2017).
Perakakis, N. et al. Physiology and role of irisin in glucose homeostasis. Nat. Rev. Endocrinol. 13, 324–337 (2017).
Google Scholar
Bao, J. F., She, Q. Y., Hu, P. P., Jia, N. & Li, A. Irisin, a fascinating field in our times. Trends Endocrinol. Metab. 33, 601–613 (2022).
Google Scholar
Mu, A. et al. Irisin acts through its integrin receptor in a two-step process involving extracellular Hsp90α. Mol. Cell 83, 1903–1920.e12 (2023).
Google Scholar
Wang, J. et al. Irisin reprograms microglia through activation of STAT6 and prevents cognitive dysfunction after surgery in mice. Brain Behav. Immun. 125, 68–91 (2025).
Google Scholar
Wang, S. et al. Integrin αvβ5 internalizes Zika virus during neural stem cells infection and provides a promising target for antiviral therapy. Cell Rep. 30, 969–983.e4 (2020).
Google Scholar
Lourenco, M. V. et al. Exercise-linked FNDC5/irisin rescues synaptic plasticity and memory defects in Alzheimer’s models. Nat. Med. 25, 165–175 (2019).
Google Scholar
Lai, W. et al. Irisin ameliorates diabetic kidney disease by restoring autophagy in podocytes. FASEB J. 37, e23175 (2023).
Google Scholar
Bzoma, B. et al. Increased circulating irisin levels in kidney transplant patients: is there a connection with glycaemic control? Int. J. Mol. Sci. (2024).
Sanchez-Tocino, M. L. et al. Sarcopenia assessed by 4-step EWGSOP2 in elderly hemodialysis patients: feasibility and limitations. PLoS ONE 17, e0261459 (2022).
Google Scholar
Hou, Y. C. et al. Indoxyl sulfate induced frailty in patients with end-stage renal disease by disrupting the PGC-1α-FNDC5 axis. Aging 15, 11532–11545 (2023).
Google Scholar
Wrann, C. D. et al. Exercise induces hippocampal BDNF through a PGC-1α/FNDC5 pathway. Cell Metab. 18, 649–659 (2013).
Google Scholar
Fontecha-Barriuso, M. et al. PGC-1α deficiency causes spontaneous kidney inflammation and increases the severity of nephrotoxic AKI. J. Pathol. 249, 65–78 (2019).
Google Scholar
Pan, H., Li, J., Zhou, Q., Zhu, F. & He, S. Protective effects of PGC-1α on the blood brain barrier after acute kidney injury. Neurochem. Res. 45, 1086–1096 (2020).
Google Scholar
Belviranli, M. & Okudan, N. Exercise training protects against aging-induced cognitive dysfunction via activation of the hippocampal PGC-1α/FNDC5/BDNF pathway. Neuromol. Med. 20, 386–400 (2018).
Google Scholar
Islam, M. R. et al. Exercise hormone irisin is a critical regulator of cognitive function. Nat. Metab. 3, 1058–1070 (2021).
Google Scholar
Kam, T. I. et al. Amelioration of pathologic α-synuclein-induced Parkinson’s disease by irisin. Proc. Natl Acad. Sci. USA 119, e2204835119 (2022).
Google Scholar
Kim, E. et al. Irisin reduces amyloid-β by inducing the release of neprilysin from astrocytes following downregulation of ERK-STAT3 signaling. Neuron 111, 3619–3633.e8 (2023).
Google Scholar
Li, X. et al. Four anti-aging drugs and calorie-restricted diet produce parallel effects in fat, brain, muscle, macrophages, and plasma of young mice. Geroscience 45, 2495–2510 (2023).
Google Scholar
Hager, M. et al. Recapitulation of anti-aging phenotypes by global overexpression of PTEN in mice. Geroscience 46, 2653–2670 (2024).
Google Scholar
Christakos, S., Dhawan, P., Verstuyf, A., Verlinden, L. & Carmeliet, G. Vitamin D: metabolism, molecular mechanism of action, and pleiotropic effects. Physiological Rev. 96, 365–408 (2016).
Google Scholar
Kagi, L. et al. Regulation of vitamin D metabolizing enzymes in murine renal and extrarenal tissues by dietary phosphate, FGF23, and 1,25(OH)2D3. PLoS ONE 13, e0195427 (2018).
Google Scholar
Pavik, I. et al. Secreted Klotho and FGF23 in chronic kidney disease stage 1 to 5: a sequence suggested from a cross-sectional study. Nephrol. Dial. Transpl. 28, 352–359 (2013).
Google Scholar
Balabanova, S. et al. 25-Hydroxyvitamin D, 24,25-dihydroxyvitamin D and 1,25-dihydroxyvitamin D in human cerebrospinal fluid. Klin. Wochenschr. 62, 1086–1090 (1984).
Google Scholar
Pardridge, W. M., Sakiyama, R. & Coty, W. A. Restricted transport of vitamin D and A derivatives through the rat blood–brain barrier. J. Neurochem. 44, 1138–1141 (1985).
Google Scholar
Eyles, D. W., Smith, S., Kinobe, R., Hewison, M. & McGrath, J. J. Distribution of the vitamin D receptor and 1α-hydroxylase in human brain. J. Chem. Neuroanat. 29, 21–30 (2005).
Google Scholar
Prufer, K., Veenstra, T. D., Jirikowski, G. F. & Kumar, R. Distribution of 1,25-dihydroxyvitamin D3 receptor immunoreactivity in the rat brain and spinal cord. J. Chem. Neuroanat. 16, 135–145 (1999).
Google Scholar
Taniura, H. et al. Chronic vitamin D3 treatment protects against neurotoxicity by glutamate in association with upregulation of vitamin D receptor mRNA expression in cultured rat cortical neurons. J. Neurosci. Res. 83, 1179–1189 (2006).
Google Scholar
Harms, L. R., Burne, T. H., Eyles, D. W. & McGrath, J. J. Vitamin D and the brain. Best. Pract. Res. Clin. Endocrinol. Metab. 25, 657–669 (2011).
Google Scholar
Sadeghian, N., Shadman, J., Moradi, A., Ghasem Golmohammadi, M. & Panahpour, H. Calcitriol protects the blood–brain barrier integrity against ischemic stroke and reduces vasogenic brain edema via antioxidant and antiapoptotic actions in rats. Brain Res. Bull. 150, 281–289 (2019).
Google Scholar
Nissou, M. F. et al. Additional clues for a protective role of vitamin D in neurodegenerative diseases: 1,25-dihydroxyvitamin D3 triggers an anti-inflammatory response in brain pericytes. J. Alzheimers Dis. 42, 789–799 (2014).
Google Scholar
Kim, H. et al. Brain endothelial P-glycoprotein level is reduced in Parkinson’s disease via a vitamin D receptor-dependent pathway. Int. J. Mol. Sci. (2020).
Liang, Q. et al. Postnatal vitamin D intake modulates hippocampal learning and memory in adult mice. Front. Neurosci. 12, 141 (2018).
Google Scholar
Latimer, C. S. et al. Vitamin D prevents cognitive decline and enhances hippocampal synaptic function in aging rats. Proc. Natl Acad. Sci. USA 111, E4359–E4366 (2014).
Google Scholar
Magdy, A. et al. Neuroprotective and therapeutic effects of calcitriol in rotenone-induced Parkinson’s disease rat model. Front. Cell Neurosci. 16, 967813 (2022).
Google Scholar
da Costa, R. O. et al. Vitamin D (VD3) intensifies the effects of exercise and prevents alterations of behavior, brain oxidative stress, and neuroinflammation, in hemiparkinsonian rats. Neurochem. Res. 48, 142–160 (2023).
Google Scholar
Keisala, T. et al. Premature aging in vitamin D receptor mutant mice. J. Steroid Biochem. Mol. Biol. 115, 91–97 (2009).
Google Scholar
Kosakai, A. et al. Degeneration of mesencephalic dopaminergic neurons in klotho mouse related to vitamin D exposure. Brain Res. 1382, 109–117 (2011).
Google Scholar
Leibrock, C. B., Voelkl, J., Kuro, O. M., Lang, F. & Lang, U. E. 1,25(OH)2D3 dependent overt hyperactivity phenotype in klotho-hypomorphic mice. Sci. Rep. 6, 24879 (2016).
Google Scholar
Du, Y. et al. Associations of vitamin D receptor polymorphisms with risk of Alzheimer’s disease, Parkinson’s disease, and mild cognitive impairment: a systematic review and meta-analysis. Front. Aging Neurosci. 16, 1377058 (2024).
Google Scholar
Zhou, C. et al. Serum 25-hydroxyvitamin D, vitamin D receptor, and vitamin-D-binding protein gene polymorphisms and risk of dementia among older adults with prediabetes. J. Gerontol. A Biol. Sci. Med. Sci. (2024).
Liu, N. et al. Vitamin D receptor gene polymorphisms and risk of Alzheimer disease and mild cognitive impairment: a systematic review and meta-analysis. Adv. Nutr. 12, 2255–2264 (2021).
Google Scholar
Homann, C. N. & Homann, B. The role of vitamin D in basal ganglia diseases. J. Integr. Neurosci. 21, 155 (2022).
Google Scholar
Wu, H. et al. Correlation between serum 25(OH)D and cognitive impairment in Parkinson’s disease. J. Clin. Neurosci. 100, 192–195 (2022).
Google Scholar
van Soest, A. P. M. et al. Concurrent nutrient deficiencies are associated with dementia incidence. Alzheimers Dement. 20, 4594–4601 (2024).
Google Scholar
Zhang, X. X. et al. Association of vitamin D levels with risk of cognitive impairment and dementia: a systematic review and meta-analysis of prospective studies. J. Alzheimers Dis. 98, 373–385 (2024).
Google Scholar
Hendriks, S. et al. Risk factors for young-onset dementia in the UK biobank. JAMA Neurol. 81, 134–142 (2024).
Google Scholar
Ghahremani, M. et al. Vitamin D supplementation and incident dementia: effects of sex, APOE, and baseline cognitive status. Alzheimers Dement 15, e12404 (2023).
Ilboudo, Y. et al. Vitamin D, cognition, and Alzheimer’s disease: observational and two-sample mendelian randomization studies. J. Alzheimers Dis. 99, 1243–1260 (2024).
Google Scholar
Mowe, M., Bohmer, T. & Haug, E. Serum calcidiol and calcitriol concentrations in elderly people: variations with age, sex, season and disease. Clin. Nutr. 15, 201–206 (1996).
Google Scholar
Azimzadeh, J. M., Shidfar, F., Jazayeri, S., Hosseini, A. F. & Ranjbaran, F. Effect of vitamin D supplementation on klotho protein, antioxidant status and nitric oxide in the elderly: a randomized, double-blinded, placebo-controlled clinical trial. Eur. J. Integr. Med. 35, 101089 (2020).
Google Scholar
Neyra, J. A. et al. Performance of soluble Klotho assays in clinical samples of kidney disease. Clin. Kidney J. 13, 235–244 (2020).
Google Scholar
Hryszko, T. et al. Cholecalciferol supplementation reduces soluble Klotho concentration in hemodialysis patients. Pol. Arch. Med. Wewn. 123, 277–281 (2013).
Google Scholar
Etemadi, J. et al. The effects of cholecalciferol supplementation on FGF23 and α-klotho in hemodialysis patients with hypovitaminosis D: a randomized, double-blind, placebo-controlled trial. J. Ren. Nutr. 32, 334–340 (2022).
Google Scholar
Lau, W. L. et al. Vitamin D receptor agonists increase klotho and osteopontin while decreasing aortic calcification in mice with chronic kidney disease fed a high phosphate diet. Kidney Int. 82, 1261–1270 (2012).
Google Scholar
Ritter, C. S., Zhang, S., Delmez, J., Finch, J. L. & Slatopolsky, E. Differential expression and regulation of Klotho by paricalcitol in the kidney, parathyroid, and aorta of uremic rats. Kidney Int. 87, 1141–1152 (2015).
Google Scholar
Montero-Odasso, M. et al. Effects of exercise alone or combined with cognitive training and vitamin D supplementation to improve cognition in adults with mild cognitive impairment: a randomized clinical trial. JAMA Netw. Open. 6, e2324465 (2023).
Google Scholar
Zammit, A. R., Katz, M. J., Bitzer, M. & Lipton, R. B. Cognitive impairment and dementia in older adults with chronic kidney disease: a review. Alzheimer Dis. Assoc. Disord. 30, 357–366 (2016).
Google Scholar
Bobot, M., Bruno, J. & Burtey, S. Cognitive impairment phenotypes in patients with CKD stages 3b and 4. Kidney360 5, 787–788 (2024).
Google Scholar
Kim, D. S., Kim, S. W. & Gil, H. W. Emotional and cognitive changes in chronic kidney disease. Korean J. Intern. Med. 37, 489–501 (2022).
Google Scholar
Wei, P., Keller, C. & Li, L. Neuropeptides in gut–brain axis and their influence on host immunity and stress. Comput. Struct. Biotechnol. J. 18, 843–851 (2020).
Google Scholar
Nahvi, R. J. & Sabban, E. L. Sex differences in the neuropeptide Y system and implications for stress related disorders. Biomolecules (2020).
Zoccali, C. et al. Neuropeptide Y as a risk factor for cardiorenal disease and cognitive dysfunction in chronic kidney disease: translational opportunities and challenges. Nephrol. Dial. Transpl. 37, ii14–ii23 (2021).
Google Scholar
Spoto, B. et al. Neuropeptide Y gene polymorphisms and chronic kidney disease progression. J. Hypertens. 42, 267–273 (2024).
Google Scholar
Sawynok, J. & Dawborn, J. K. Plasma concentration and urinary excretion of guanidine derivatives in normal subjects and patients with renal failure. Clin. Exp. Pharmacol. Physiol. 2, 1–15 (1975).
Google Scholar
Berlinck, R. G. S., Bertonha, A. F., Takaki, M. & Rodriguez, J. P. G. The chemistry and biology of guanidine natural products. Nat. Prod. Rep. 34, 1264–1301 (2017).
Google Scholar
Salminen, A. Activation of aryl hydrocarbon receptor (AhR) in Alzheimer’s disease: role of tryptophan metabolites generated by gut host-microbiota. J. Mol. Med. 101, 201–222 (2023).
Google Scholar
Tsuji, K. et al. The gut–kidney axis in chronic kidney diseases. Diagnostics (2024).
Turski, W. A. et al. On the toxicity of kynurenic acid in vivo and in vitro. Pharmacol. Rep. 66, 1127–1133 (2014).
Google Scholar
Stringuetta Belik, F. et al. Influence of intradialytic aerobic training in cerebral blood flow and cognitive function in patients with chronic kidney disease: a pilot randomized controlled trial. Nephron 140, 9–17 (2018).
Google Scholar
McAdams-DeMarco, M. A. et al. Intradialytic cognitive and exercise training may preserve cognitive function. Kidney Int. Rep. 3, 81–88 (2018).
Google Scholar
Otobe, Y. et al. Physical exercise improves cognitive function in older adults with stage 3-4 chronic kidney disease: a randomized controlled trial. Am. J. Nephrol. 52, 929–939 (2021).
Google Scholar
Bernier-Jean, A. et al. Exercise training for adults undergoing maintenance dialysis. Cochrane Database Syst. Rev. 1, CD014653 (2022).
Google Scholar
Bradshaw, E., Alejmi, A., Rossetti, G., D’Avossa, G. & Macdonald, J. H. Exercise and cognitive function in chronic kidney disease: a systematic review and meta-analysis of efficacy and harms. Clin. J. Am. Soc. Nephrol. 19, 1461–1472 (2024).
Google Scholar
Rose, M. et al. The CONVINCE randomized trial found positive effects on quality of life for patients with chronic kidney disease treated with hemodiafiltration. Kidney Int. 106, 961–971 (2024).
Google Scholar
link










More Stories
Russia turns to high altitude balloons after Starlink restrictions disrupt battlefield communications across Russian-controlled territories
What CES 2026 didn’t show: The quiet crisis in wireless capacity nobody is talking about
Recent AWS issues blamed on AI tools – at least two incidents affected some Amazon services