Standard protocol approvals, registrations, and patient consents
An overview of the study design is presented in Fig. 1. This study was approved by the Ethics Committee of Beijing Tiantan Hospital, Capital Medical University (approval number: KY2022-044), as a sub-study of the ongoing China HeadAche DIsorders RegiStry Study (CHAIRS; NCT05334927). Written informed consent was obtained from all participants in accordance with the Declaration of Helsinki.
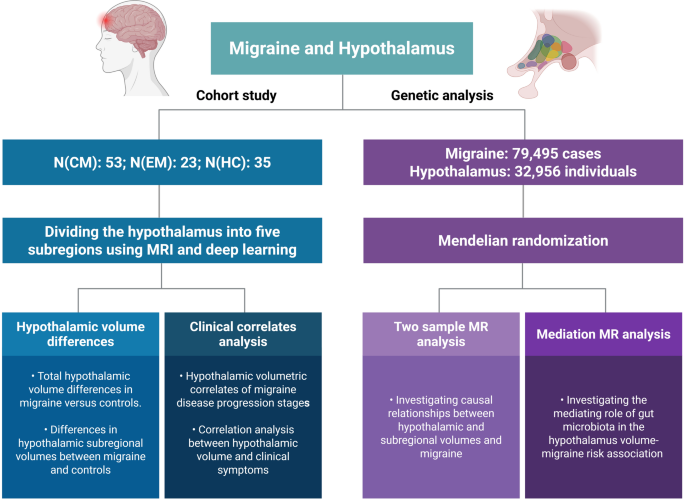
Study design flowchart. Deep learning-based MRI segmentation of the hypothalamus combined with Mendelian randomization analysis provides imaging and genetic evidence for the impacts of the hypothalamus on migraines. Abbreviations: CM, chronic migraine; EM, episiod migraine; HC, healthy control
Participants for neuroimaging analysis
This observational, cross-sectional study included 111 participants recruited consecutively from the Headache Outpatient Unit of Beijing Tiantan Hospital, Capital Medical University, between October 2020 and March 2023. The cohort comprised 53 individuals with chronic migraine (CM), 23 with episodic migraine (EM), and 35 HCs. All migraine diagnoses were made according to the International Classification of Headache Disorders, 3rd edition (ICHD-3) [2]. Eligible participants were aged 14 to 60 years, capable of completing MRI, and had not received preventive treatment in the preceding three months. There was no acute medication the day before the MRI scan. General exclusion criteria for both patients and HC included the presence of other primary headache or pain syndromes, pregnancy or lactation, neurological, cardiovascular, or endocrine disorders, substance abuse, a first-degree relative with a headache disorder, poor-quality MRI scans (e.g., artefacts or incomplete data).
Demographic assessment
Data collected from all patients included demographics, age (years), body mass index (BMI), gender (female/male), attack frequency (days/month), headache duration (years), Visual Analogue Scale (VAS) scores. Headache-related disability was quantified using the Headache Impact Test-6 (HIT-6).
Hypothalamic imaging acquisition and segmentation
High-resolution 3D T1-weighted structural MRI data were acquired at the National Neurological Center of Beijing Tiantan Hospital using a 3.0 Tesla GE Signa Premier scanner (GE Healthcare) equipped with a 48-channel head coil. Participants were instructed to remain still with their eyes closed throughout the scan. Imaging parameters for the magnetization-prepared rapid gradient echo (MP-RAGE) sequence included: preparation time = 880 ms, acquisition time = 4 min, recovery time = 400 ms, field of view = 250 × 250 mm2, acceleration factor = 2, flip angle = 8°, 192 sagittal slices, and isotropic spatial resolution of 1 × 1 × 1 mm3.
Volumes of the hypothalamus and its five subregions—including the anterior superior (a-sHyp), anterior inferior (a-iHyp), superior tuberal (supTub), inferior tuberal (infTub), and posterior (posHyp) subunits—were delineated using a fully automated segmentation tool [16]. This tool is based on a deep convolutional neural network trained on T1-weighted MRI scans from 37 manually labeled subjects. To artificially increase the number of training samples and avoid preprocessing, the MRI scans were first subjected to data augmentation. The network adopts a 3D U-Net architecture, featuring a symmetric encoder–decoder structure with skip connections. By processing the full 3D context of input scans, it preserves spatial continuity across slices, enabling more accurate delineation of complex anatomical structures. Its symmetric encoder–decoder design with skip connections allows for precise localization by integrating both low-level spatial detail and high-level semantic information. Moreover, 3D U-Net supports end-to-end learning, performs effectively on small datasets with appropriate data augmentation, and can be flexibly applied to multiclass segmentation tasks. These characteristics make it particularly well-suited for robust and reproducible segmentation in brain MRI analyses. This method demonstrated high reproducibility and did not require additional preprocessing. After segmenting the hypothalamus into five subregions, the total hypothalamic volume was calculated as the sum of the volumes of these subregions.
Statistical analysis for demographic, clinical and neuroimaging data
Statistical analyses were performed using IBM SPSS Statistics 27.0. During the comparison of demographic characteristics, continuous variables were assessed for normality using the Shapiro–Wilk test. Normally distributed data are presented as mean ± standard deviation (SD), while non-normally distributed data are presented as median with interquartile range (IQR). Categorical variables are summarized as frequencies and percentages. Group comparisons between two groups were conducted using independent samples t-tests for normally distributed variables and Mann-Whitney U tests for non-normally distributed variables. Categorical variables were analyzed using Pearson’s χ² test or the continuity-corrected χ² test where appropriate. For comparisons among three groups (HC, EM, and CM), one-way analysis of variance (ANOVA) was used for normally distributed continuous variables, and the Kruskal–Wallis test was used when variables violated normality assumptions.
For comparisons of hypothalamic volumes between the HC and migraine groups, linear regression models were used, adjusting for age, gender, BMI, and total intracranial volume (TIV). For comparisons of hypothalamic volumes across clinical stage of migraine (HC to EM to CM), analysis of covariance (ANCOVA) was conducted, adjusting for age, gender, BMI, and TIV. Type III sum of squares was used to account for unbalanced design. Post hoc pairwise comparisons were conducted using estimated marginal means (emmeans) with FDR correction. Pearson correlation analyses performed between significantly different volumetric measures and clinical scores in the patient group.
GWAS of the hypothalamus and migraine
GWAS summary statistics for the volume of the hypothalamus and its five subregions were obtained from a previous study involving MRI and genetic data from 32,956 individuals [17]. This study employed the same segmentation procedure as the present study and performed GWAS, identifying 23 loci associated with hypothalamic volume, which were enriched for genes involved in intracellular trafficking and steroid metabolism.
GWAS smmary statistics for migraine were obtained from the largest GWAS to date (Ncase = 79,495, Ncontrol = 1,259,808), which also provided subtype-specific data for migraine with aura (MA; Ncase = 16,603, Ncontrol = 1,336,517) and without aura (MO; Ncase = 11,718, Ncontrol = 1,330,747) [18]. These participants were recruited from one tertiary headache clinics (N = 280,844) and five population-based cohorts (N = 1,058,459) through various methods, such as advertisements, the project’s website, national media campaigns, and referrals from headache centers. Detailed recruitment information is available in the respective cohort descriptions. All GWAS included in the present study were exclusively conducted among participants of European ancestry.
Mendelian randomization analysis
We used the TwoSampleMR package ( to evaluate bidirectional causal relationships between the volumes of hypothalamic regions identified as significant in imaging analyses and the risk of migraine and its subtypes (MO and MA). The forward MR analysis was performed with hypothalamic volume as exposure and migraine as outcome. Conversely, the reverse MR analysis was performed with migraine as exposure and hypothalamic volume as outcome. Genetic instrumental variables (IVs) were defined as genome-wide significant (p < 5 × 10−8) Single Nucleotide Polymorphisms (SNPs) with a minor allele frequency (MAF) > 0.01. To reduce bias from weak instruments, only SNPs with F-statistics > 10 were retained [19, 20]. The resulting genetic IVs were pruned to high independence with a r2 threshold of 0.001 and a window size of 10 Mb. To improve instrument quality, heterogeneity and outliers were assessed using ivw_radial and egger_radial functions in RadialMR (v0.4, applying Q and Q′ statistics at a nominal significance level of 0.05. Ambiguous or palindromic variants were corrected or excluded during harmonization to ensure allele alignment.
Causal estimates were obtained using the inverse variance-weighted (IVW) method, with a random-effects model for traits with more than three instruments and a fixed-effects model for those with two or three [21, 22]. Where only one SNP was available, the Wald ratio was applied [23]. Odds ratios (ORs) for disease risk were reported per standard deviation (SD) increase in hypothalamic volume. FDR correction was applied to address multiple comparisons.
Sensitivity analyses were performed to assess potential pleiotropy and heterogeneity. Horizontal pleiotropy was evaluated using MR-Egger regression and the MR-PRESSO Global test [24, 25], while Cochran’s Q statistic was used to assess heterogeneity among instrumental variables [26].
Mediation analysis of hypothalamus, gut microbiome, and migraine
Given the emerging role of the gut microbiome in migraine pathophysiology [11, 27] and recent evidence linking hypothalamic structure to gut microbial composition [28], we conducted a two-step MR mediation analysis to investigate whether gut microbiota mediate the relationship between hypothalamic volume and migraine risk. Microbiome GWAS summary statistics were obtained from the MiBioGen consortium [29]. This study analyzed 16 S rRNA gene sequencing and genotype data from 18,340 individuals across 24 cohorts in multiple countries, identifying associations between host genetic variation and gut microbial taxa. The dataset included 211 taxa across 131 genera, 35 families, 20 orders, 16 classes, and 9 phyla [29].
First, we assessed the causal effect of hypothalamic volume on gut microbial taxa using MR. We applied FDR correction to account for multiple testing. Second, taxa showing significant associations were tested for causal effects on migraine. Due to the limited number of genome-wide significant variants for microbial traits, we used a locus-wide significance threshold (p < 1.0 × 10−5) for instrument selection.
Mediation was quantified using the “product of coefficients” method [30], where the indirect effect was calculated as the product of the MR estimates from hypothalamic volume to microbiota and from microbiota to migraine. Standard errors were computed using the delta method [31], and the proportion mediated was defined as the ratio of the indirect effect to the total effect. A p-value < 0.05 was considered statistically significant for mediation testing.
link











More Stories
What modern medicine gets wrong about body’s operating system
Choosing birth control? Weighing the risks and benefits
An indie brand’s approach to hormone-conscious skincare