Ying, M. et al. Disease burden and epidemiological trends of chronic kidney disease at the global, regional, national levels from 1990 to 2019. Nephron 148, 113–123 (2024).
Google Scholar
Kovesdy, C. P. Epidemiology of chronic kidney disease: an update 2022. Kidney Int. Suppl. 12, 7–11 (2022).
Google Scholar
International Diabetes Federation. Diabetes Atlas 10th edn (International Diabetes Federation, 2021).
Swinburn, B. A. et al. The global syndemic of obesity, undernutrition, and climate change: the Lancet Commission report. Lancet 393, 791–846 (2019).
Google Scholar
World Health Organization (WHO). Obesity and Overweight. (2025).
Cameron, N. A. et al. Quantifying the sex-race/ethnicity-specific burden of obesity on incident diabetes mellitus in the United States, 2001 to 2016: MESA and NHANES. J. Am. Heart Assoc. 10, e018799 (2021).
Google Scholar
Murray, C. J. L. et al. Global burden of 87 risk factors in 204 countries and territories, 1990–2019: a systematic analysis for the Global Burden of Disease study 2019. Lancet 396, 1223–1249 (2020).
Google Scholar
Institute for Health Metrics and Evaluation (IHME). Global Burden of Disease Study 2019 (GBD 2019) Covariates 1980–2019, https://ghdx.healthdata.org/record/global-burden-disease-study-2019-gbd-2019-covariates-1980-2019.
Okunogbe, A. et al. Economic impacts of overweight and obesity: current and future estimates for 161 countries. BMJ Glob. Health 7, e009773 (2022).
Google Scholar
Polyzos, S. A. & Mantzoros, C. S. Obesity: seize the day, fight the fat. Metab. Clin. Exp. 92, 1–5 (2019).
Google Scholar
Boutari, C. & Mantzoros, C. S. A 2022 update on the epidemiology of obesity and a call to action: as its twin COVID-19 pandemic appears to be receding, the obesity and dysmetabolism pandemic continues to rage on. Metab. Clin. Exp. 133, 155217 (2022).
Google Scholar
Tuttle K. R. et al. Risks of kidney failure and death in a real-world population with diabetes. Clin. J. Am. Soc. Nephrol. (2025).
Grams, M. E. et al. Estimated glomerular filtration rate, albuminuria, and adverse outcomes: an individual-participant data meta-analysis. JAMA 330, 1266–1277 (2023).
Google Scholar
Jankowski, J., Floege, J., Fliser, D., Bohm, M. & Marx, N. Cardiovascular disease in chronic kidney disease: pathophysiological insights and therapeutic options. Circulation 143, 1157–1172 (2021).
Google Scholar
Li, X. & Lindholm, B. Cardiovascular risk prediction in chronic kidney disease. Am. J. Nephrol. 53, 730–739 (2022).
Google Scholar
Ndumele, C. E. et al. Cardiovascular-kidney-metabolic health: a presidential advisory from the American Heart Association. Circulation 148, 1606–1635 (2023).
Google Scholar
Perkovic, V. et al. Effects of semaglutide on chronic kidney disease in patients with type 2 diabetes. N. Engl. J. Med. 391, 109–212 (2024).
Google Scholar
Parker, V. E. R. et al. Efficacy and safety of cotadutide, a dual glucagon-like peptide-1 and glucagon receptor agonist, in a randomized phase 2a study of patients with type 2 diabetes and chronic kidney disease. Diabetes Obes. Metab. 24, 1360–1369 (2022).
Google Scholar
Nauck, M. A. et al. Preserved incretin activity of glucagon-like peptide 1 [7-36 amide] but not of synthetic human gastric inhibitory polypeptide in patients with type-2 diabetes mellitus. J. Clin. Invest. 91, 301–307 (1993).
Google Scholar
Kreymann, B., Williams, G., Ghatei, M. A. & Bloom, S. R. Glucagon-like peptide-1 7-36: a physiological incretin in man. Lancet 2, 1300–1304 (1987).
Google Scholar
Bagger, J. I. et al. Impaired regulation of the incretin effect in patients with type 2 diabetes. J. Clin. Endocrinol. Metab. 96, 737–745 (2011).
Google Scholar
Pederson, R. A. & McIntosh, C. H. Discovery of gastric inhibitory polypeptide and its subsequent fate: personal reflections. J. Diabetes Investig. 7, 4–7 (2016).
Google Scholar
Nauck, M. A., Bartels, E., Orskov, C., Ebert, R. & Creutzfeldt, W. Additive insulinotropic effects of exogenous synthetic human gastric inhibitory polypeptide and glucagon-like peptide-1-(7-36) amide infused at near-physiological insulinotropic hormone and glucose concentrations. J. Clin. Endocrinol. Metab. 76, 912–917 (1993).
Google Scholar
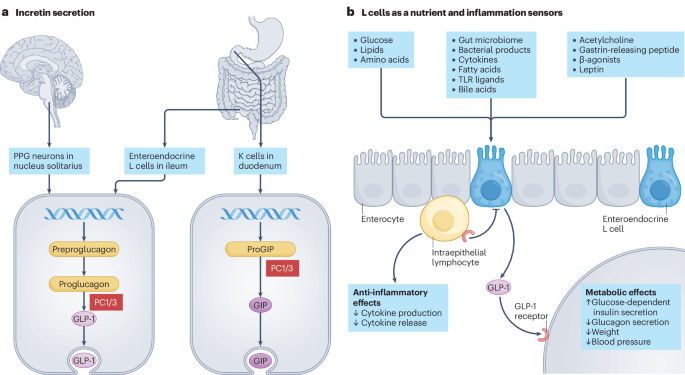
Holst, J. J., Albrechtsen, N. J. W., Rosenkilde, M. M. & Deacon, C. F. Physiology of the incretin hormones, GIP and GLP-1-regulation of release and posttranslational modifications. Compr. Physiol. 9, 1339–1381 (2019).
Google Scholar
Alicic, R. Z., Cox, E. J., Neumiller, J. J. & Tuttle, K. R. Incretin drugs in diabetic kidney disease: biological mechanisms and clinical evidence. Nat. Rev. Nephrol. 17, 227–244 (2021).
Google Scholar
Holt, M. K. et al. Preproglucagon neurons in the nucleus of the solitary tract are the main source of brain GLP-1, mediate stress-induced hypophagia, and limit unusually large intakes of food. Diabetes 68, 21–33 (2019).
Google Scholar
Nauck, M. A., Quast, D. R., Wefers, J. & Pfeiffer, A. F. H. The evolving story of incretins (GIP and GLP-1) in metabolic and cardiovascular disease: a pathophysiological update. Diabetes Obes. Metab. 23, 5–29 (2021).
Google Scholar
Gunawardene, A. R., Corfe, B. M. & Staton, C. A. Classification and functions of enteroendocrine cells of the lower gastrointestinal tract. Int. J. Exp. Pathol. 92, 219–231 (2011).
Google Scholar
Jin, S. L. et al. Distribution of glucagonlike peptide I (GLP-I), glucagon, and glicentin in the rat brain: an immunocytochemical study. J. Comp. Neurol. 271, 519–532 (1988).
Google Scholar
Morrow, N. M., Morissette, A. & Mulvihill, E. E. Immunomodulation and inflammation: Role of GLP-1R and GIPR expressing cells within the gut. Peptides 176, 171200 (2024).
Google Scholar
Capozzi, M. E., D’Alessio, D. A. & Campbell, J. E. The past, present, and future physiology and pharmacology of glucagon. Cell Metab. 34, 1654–1674 (2022).
Google Scholar
Deacon, C. F. What do we know about the secretion and degradation of incretin hormones? Regul. Pept. 128, 117–124 (2005).
Google Scholar
Drucker, D. J. Mechanisms of action and therapeutic application of glucagon-like peptide-1. Cell Metab. 27, 740–756 (2018).
Google Scholar
Balks, H. J., Holst, J. J., von zur Muhlen, A. & Brabant, G. Rapid oscillations in plasma glucagon-like peptide-1 (GLP-1) in humans: cholinergic control of GLP-1 secretion via muscarinic receptors. J. Clin. Endocrinol. Metab. 82, 786–790 (1997).
Google Scholar
Muller, T. D. et al. Glucagon-like peptide 1 (GLP-1). Mol. Metab. 30, 72–130 (2019).
Google Scholar
Grasset, E. et al. A specific gut microbiota dysbiosis of type 2 diabetic mice induces GLP-1 resistance through an enteric no-dependent and gut-brain axis mechanism. Cell Metab. 26, 278 (2017).
Google Scholar
Ellingsgaard, H. et al. Interleukin-6 enhances insulin secretion by increasing glucagon-like peptide-1 secretion from L cells and alpha cells. Nat. Med. 17, 1481–1489 (2011).
Google Scholar
Lebherz, C. et al. Interleukin-6 predicts inflammation-induced increase of glucagon-like peptide-1 in humans in response to cardiac surgery with association to parameters of glucose metabolism. Cardiovasc. Diabetol. 15, 21 (2016).
Google Scholar
Kahles, F. et al. GLP-1 secretion is increased by inflammatory stimuli in an IL-6-dependent manner, leading to hyperinsulinemia and blood glucose lowering. Diabetes 63, 3221–3229 (2014).
Google Scholar
Kahles, F. et al. Glucose-dependent insulinotropic peptide secretion is induced by inflammatory stimuli in an interleukin-1-dependent manner in mice. Diabetes Obes. Metab. 18, 1147–1151 (2016).
Google Scholar
Lebrun, L. J. et al. Enteroendocrine L cells sense LPS after gut barrier injury to enhance GLP-1 secretion. Cell Rep. 21, 1160–1168 (2017).
Google Scholar
Yamada, Y. et al. Human gastric inhibitory polypeptide receptor: cloning of the gene (GIPR) and cDNA. Genomics 29, 773–776 (1995).
Google Scholar
Thorens, B. Expression cloning of the pancreatic beta cell receptor for the gluco-incretin hormone glucagon-like peptide 1. Proc. Natl Acad. Sci. USA 89, 8641–8645 (1992).
Google Scholar
Zheng, Z. et al. Glucagon-like peptide-1 receptor: mechanisms and advances in therapy. Signal. Transduct. Target. Ther. 9, 234 (2024).
Google Scholar
Zhang, L., Zhang, W. & Tian, X. The pleiotropic of GLP-1/GLP-1R axis in central nervous system diseases. Int. J. Neurosci. 133, 473–491 (2023).
Google Scholar
Meyer-Gerspach, A. C. et al. Endogenous GLP-1 alters postprandial functional connectivity between homeostatic and reward-related brain regions involved in regulation of appetite in healthy lean males: a pilot study. Diabetes Obes. Metab. 20, 2330–2338 (2018).
Google Scholar
Ansari, S., Khoo, B. & Tan, T. Targeting the incretin system in obesity and type 2 diabetes mellitus. Nat. Rev. Endocrinol. 20, 447–459 (2024).
Google Scholar
Sun, E. W. L., Martin, A. M., Young, R. L. & Keating, D. J. The regulation of peripheral metabolism by gut-derived hormones. Front. Endocrinol. 9, 754 (2018).
Google Scholar
Llewellyn-Smith, I. J., Reimann, F., Gribble, F. M. & Trapp, S. Preproglucagon neurons project widely to autonomic control areas in the mouse brain. Neuroscience 180, 111–121 (2011).
Google Scholar
Drucker, D. J. & Holst, J. J. The expanding incretin universe: from basic biology to clinical translation. Diabetologia 66, 1765–1779 (2023).
Google Scholar
Frias, J. P. et al. Tirzepatide versus semaglutide once weekly in patients with type 2 diabetes. N. Engl. J. Med. 385, 503–515 (2021).
Google Scholar
Jastreboff, A. M. et al. Triple-hormone-receptor agonist retatrutide for obesity — a phase 2 trial. N. Engl. J. Med. 389, 514–526 (2023).
Google Scholar
Pyke, C. et al. GLP-1 receptor localization in monkey and human tissue: novel distribution revealed with extensively validated monoclonal antibody. Endocrinology 155, 1280–1290 (2014).
Google Scholar
Schlatter, P., Beglinger, C., Drewe, J. & Gutmann, H. Glucagon-like peptide 1 receptor expression in primary porcine proximal tubular cells. Regul. Pept. 141, 120–128 (2007).
Google Scholar
Korner, M., Stockli, M., Waser, B. & Reubi, J. C. GLP-1 receptor expression in human tumors and human normal tissues: potential for in vivo targeting. J. Nucl. Med. 48, 736–743 (2007).
Google Scholar
Crajoinas, R. O. et al. Mechanisms mediating the diuretic and natriuretic actions of the incretin hormone glucagon-like peptide-1. Am. J. Physiol. Renal Physiol. 301, F355–F363 (2011).
Google Scholar
Farah, L. X. et al. The physiological role of glucagon-like peptide-1 in the regulation of renal function. Am. J. Physiol. Renal Physiol. 310, F123–F127 (2016).
Google Scholar
Gutzwiller, J. P. et al. Glucagon-like peptide 1 induces natriuresis in healthy subjects and in insulin-resistant obese men. J. Clin. Endocrinol. Metab. 89, 3055–3061 (2004).
Google Scholar
Skov, J. et al. Glucagon-like peptide-1 (GLP-1): effect on kidney hemodynamics and renin-angiotensin-aldosterone system in healthy men. J. Clin. Endocrinol. Metab. 98, E664–E671 (2013).
Google Scholar
Gutzwiller, J. P. et al. Glucagon-like peptide-1 is involved in sodium and water homeostasis in humans. Digestion 73, 142–150 (2006).
Google Scholar
Tonneijck, L. et al. Acute renal effects of the GLP-1 receptor agonist exenatide in overweight type 2 diabetes patients: a randomised, double-blind, placebo-controlled trial. Diabetologia 59, 1412–1421 (2016).
Google Scholar
Tonneijck, L. et al. Renal tubular effects of prolonged therapy with the GLP-1 receptor agonist lixisenatide in patients with type 2 diabetes mellitus. Am. J. Physiol. Renal Physiol. 316, F231–F240 (2019).
Google Scholar
Rieg, T. et al. Natriuretic effect by exendin-4, but not the DPP-4 inhibitor alogliptin, is mediated via the GLP-1 receptor and preserved in obese type 2 diabetic mice. Am. J. Physiol. Ren. Physiol. 303, F963–F971 (2012).
Google Scholar
Asmar, A. et al. The renal extraction and the natriuretic action of GLP-1 in Humans depend on interaction with the GLP-1 receptor. J. Clin. Endocrinol. Metab. 106, e11–e19 (2021).
Google Scholar
Thomson, S. C., Kashkouli, A., Liu, Z. Z. & Singh, P. Renal hemodynamic effects of glucagon-like peptide-1 agonist are mediated by nitric oxide but not prostaglandin. Am. J. Physiol. Renal Physiol. 313, F854–F858 (2017).
Google Scholar
Fonseca, V. A. et al. Reductions in systolic blood pressure with liraglutide in patients with type 2 diabetes: insights from a patient-level pooled analysis of six randomized clinical trials. J. Diabetes Complications 28, 399–405 (2014).
Google Scholar
Park, J. et al. Single-cell transcriptomics of the mouse kidney reveals potential cellular targets of kidney disease. Science 360, 758–763 (2018).
Google Scholar
Poll, B. G., Chen, L., Chou, C. L., Raghuram, V. & Knepper, M. A. Landscape of GPCR expression along the mouse nephron. Am. J. Physiol. Renal Physiol. 321, F50–F68 (2021).
Google Scholar
Limbutara, K., Chou, C. L. & Knepper, M. A. Quantitative proteomics of all 14 renal tubule segments in rat. J. Am. Soc. Nephrol. 31, 1255–1266 (2020).
Google Scholar
Takeda, Y. et al. Increment of plasma glucose by exogenous glucagon is associated with present and future renal function in type 2 diabetes: a retrospective study from glucagon stimulation test. BMC Endocr. Disord. 19, 99 (2019).
Google Scholar
Ahloulay, M., Dechaux, M., Laborde, K. & Bankir, L. Influence of glucagon on GFR and on urea and electrolyte excretion: direct and indirect effects. Am. J. Physiol. 269, F225–F235 (1995).
Google Scholar
Mercier, O. et al. Effects of glucagon on H+-HCO3– transport in Henle’s loop, distal tubule, and collecting ducts in the rat. Am. J. Physiol. 257, F1003–F1014 (1989).
Google Scholar
He, S. et al. Gut intraepithelial T cells calibrate metabolism and accelerate cardiovascular disease. Nature 566, 115–119 (2019).
Google Scholar
Wong, C. K. et al. Divergent roles for the gut intraepithelial lymphocyte GLP-1R in control of metabolism, microbiota, and T cell-induced inflammation. Cell Metab. 34, 1514–1531.e1517 (2022).
Google Scholar
Bubeck Wardenburg, J. et al. Phosphorylation of SLP-76 by the ZAP-70 protein-tyrosine kinase is required for T-cell receptor function. J. Biol. Chem. 271, 19641–19644 (1996).
Google Scholar
Mustelin, T. & Tasken, K. Positive and negative regulation of T-cell activation through kinases and phosphatases. Biochem. J. 371, 15–27 (2003).
Google Scholar
Kodera, R. et al. Glucagon-like peptide-1 receptor agonist ameliorates renal injury through its anti-inflammatory action without lowering blood glucose level in a rat model of type 1 diabetes. Diabetologia 54, 965–978 (2011).
Google Scholar
Heng, T. S., Painter, M. W. & Immunological Genome Project, C The immunological genome project: networks of gene expression in immune cells. Nat. Immunol. 9, 1091–1094 (2008).
Google Scholar
Higashijima, Y., Tanaka, T., Yamaguchi, J., Tanaka, S. & Nangaku, M. Anti-inflammatory role of DPP-4 inhibitors in a nondiabetic model of glomerular injury. Am. J. Physiol. Renal Physiol. 308, F878–F887 (2015).
Google Scholar
Mantelmacher, F. D. et al. GIP regulates inflammation and body weight by restraining myeloid-cell-derived S100A8/A9. Nat. Metab. 1, 58–69 (2019).
Google Scholar
Kahn, S. E. et al. Evidence of cosecretion of islet amyloid polypeptide and insulin by beta-cells. Diabetes 39, 634–638 (1990).
Google Scholar
Inoue, K., Hisatomi, A., Umeda, F. & Nawata, H. Effects of glucagon-like peptide 1 (7-36) amide and glucagon on amylin release from perfused rat pancreas. Horm. Metab. Res. 23, 407–409 (1991).
Google Scholar
Westermark, P., Andersson, A. & Westermark, G. T. Islet amyloid polypeptide, islet amyloid, and diabetes mellitus. Physiol. Rev. 91, 795–826 (2011).
Google Scholar
Hay, D. L., Chen, S., Lutz, T. A., Parkes, D. G. & Roth, J. D. Amylin: pharmacology, physiology, and clinical potential. Pharmacol. Rev. 67, 564–600 (2015).
Google Scholar
Cao, J. et al. A structural basis for amylin receptor phenotype. Science 375, eabm9609 (2022).
Google Scholar
Lutz, T. A. Creating the amylin story. Appetite 172, 105965 (2022).
Google Scholar
Zakariassen, H. L., John, L. M. & Lutz, T. A. Central control of energy balance by amylin and calcitonin receptor agonists and their potential for treatment of metabolic diseases. Basic. Clin. Pharmacol. Toxicol. 127, 163–177 (2020).
Google Scholar
Erzen, S., Tonin, G., Jurisic Erzen, D. & Klen, J. Amylin, another important neuroendocrine hormone for the treatment of diabesity. Int. J. Mol. Sci. 25, 1517 (2024).
Google Scholar
Coester, B., Foll, C. L. & Lutz, T. A. Viral depletion of calcitonin receptors in the area postrema: a proof-of-concept study. Physiol. Behav. 223, 112992 (2020).
Google Scholar
Lutz, T. A. Pancreatic amylin as a centrally acting satiating hormone. Curr. Drug. Targets 6, 181–189 (2005).
Google Scholar
Mack, C. M. et al. Glucoregulatory effects and prolonged duration of action of davalintide: a novel amylinomimetic peptide. Diabetes Obes. Metab. 13, 1105–1113 (2011).
Google Scholar
Potes, C. S. & Lutz, T. A. Brainstem mechanisms of amylin-induced anorexia. Physiol. Behav. 100, 511–518 (2010).
Google Scholar
Roth, J. D. et al. Leptin responsiveness restored by amylin agonism in diet-induced obesity: evidence from nonclinical and clinical studies. Proc. Natl Acad. Sci. USA 105, 7257–7262 (2008).
Google Scholar
Turek, V. F. et al. Mechanisms of amylin/leptin synergy in rodent models. Endocrinology 151, 143–152 (2010).
Google Scholar
Liberini, C. G. et al. Amylin receptor components and the leptin receptor are co-expressed in single rat area postrema neurons. Eur. J. Neurosci. 43, 653–661 (2016).
Google Scholar
Le Foll, C. et al. Amylin-induced central IL-6 production enhances ventromedial hypothalamic leptin signaling. Diabetes 64, 1621–1631 (2015).
Google Scholar
Ozcan, U. et al. Endoplasmic reticulum stress links obesity, insulin action, and type 2 diabetes. Science 306, 457–461 (2004).
Google Scholar
Kahn, S. E., Hull, R. L. & Utzschneider, K. M. Mechanisms linking obesity to insulin resistance and type 2 diabetes. Nature 444, 840–846 (2006).
Google Scholar
Mitrofanova, A., Merscher, S. & Fornoni, A. Kidney lipid dysmetabolism and lipid droplet accumulation in chronic kidney disease. Nat. Rev. Nephrol. 19, 629–645 (2023).
Google Scholar
Saxton, S. N., Clark, B. J., Withers, S. B., Eringa, E. C. & Heagerty, A. M. Mechanistic links between obesity, diabetes, and blood pressure: role of perivascular adipose tissue. Physiol. Rev. 99, 1701–1763 (2019).
Google Scholar
Hall, J. E., do Carmo, J. M., da Silva, A. A., Wang, Z. & Hall, M. E. Obesity, kidney dysfunction and hypertension: mechanistic links. Nat. Rev. Nephrol. 15, 367–385 (2019).
Google Scholar
Brownlee, M. The pathobiology of diabetic complications: a unifying mechanism. Diabetes 54, 1615–1625 (2005).
Google Scholar
Weigert, C., Brodbeck, K., Lehmann, R., Haring, H. U. & Schleicher, E. D. Overexpression of glutamine:fructose-6-phosphate-amidotransferase induces transforming growth factor-beta1 synthesis in NIH-3T3 fibroblasts. FEBS Lett. 488, 95–99 (2001).
Google Scholar
Deb, D. K., Bao, R. & Li, Y. C. Critical role of the cAMP-PKA pathway in hyperglycemia-induced epigenetic activation of fibrogenic program in the kidney. FASEB J. 31, 2065–2075 (2017).
Google Scholar
Miller, C. G., Pozzi, A., Zent, R. & Schwarzbauer, J. E. Effects of high glucose on integrin activity and fibronectin matrix assembly by mesangial cells. Mol. Biol. Cell 25, 2342–2350 (2014).
Google Scholar
Wright, R. J. et al. Effects of acute insulin-induced hypoglycemia on indices of inflammation: putative mechanism for aggravating vascular disease in diabetes. Diabetes Care 33, 1591–1597 (2010).
Google Scholar
Ceriello, A. et al. Glucagon-like peptide 1 reduces endothelial dysfunction, inflammation, and oxidative stress induced by both hyperglycemia and hypoglycemia in type 1 diabetes. Diabetes Care 36, 2346–2350 (2013).
Google Scholar
Morcos, M. et al. Activation of tubular epithelial cells in diabetic nephropathy. Diabetes 51, 3532–3544 (2002).
Google Scholar
Wendt, T. et al. Glucose, glycation, and RAGE: implications for amplification of cellular dysfunction in diabetic nephropathy. J. Am. Soc. Nephrol. 14, 1383–1395 (2003).
Google Scholar
Bierhaus, A. et al. Understanding RAGE, the receptor for advanced glycation end products. J. Mol. Med. 83, 876–886 (2005).
Google Scholar
Nagareddy, P. R. et al. Hyperglycemia promotes myelopoiesis and impairs the resolution of atherosclerosis. Cell Metab. 17, 695–708 (2013).
Google Scholar
Chen, T., Cao, Q., Wang, Y. & Harris, D. C. H. M2 macrophages in kidney disease: biology, therapies, and perspectives. Kidney Int. 95, 760–773 (2019).
Google Scholar
Chow, F. Y., Nikolic-Paterson, D. J., Ozols, E., Atkins, R. C. & Tesch, G. H. Intercellular adhesion molecule-1 deficiency is protective against nephropathy in type 2 diabetic db/db mice. J. Am. Soc. Nephrol. 16, 1711–1722 (2005).
Google Scholar
Chow, F. Y. et al. Monocyte chemoattractant protein-1-induced tissue inflammation is critical for the development of renal injury but not type 2 diabetes in obese db/db mice. Diabetologia 50, 471–480 (2007).
Google Scholar
Zeng, L. F., Xiao, Y. & Sun, L. A glimpse of the mechanisms related to renal fibrosis in diabetic nephropathy. Adv. Exp. Med. Biol. 1165, 49–79 (2019).
Google Scholar
Baggiolini, M. Chemokines and leukocyte traffic. Nature 392, 565–568 (1998).
Google Scholar
Tang, S. C. W. & Yiu, W. H. Innate immunity in diabetic kidney disease. Nat. Rev. Nephrol. 16, 206–222 (2020).
Google Scholar
Wong, C. K. et al. Central glucagon-like peptide 1 receptor activation inhibits Toll-like receptor agonist-induced inflammation. Cell Metab. 36, 130–143 e135 (2024).
Google Scholar
Yiu, W. H., Lin, M. & Tang, S. C. Toll-like receptor activation: from renal inflammation to fibrosis. Kidney Int. Suppl. 4, 20–25 (2014).
Google Scholar
Lin, M. et al. Toll-like receptor 4 promotes tubular inflammation in diabetic nephropathy. J. Am. Soc. Nephrol.23, 86–102 (2012).
Google Scholar
Huang, G., Zhang, Y., Zhang, Y. & Ma, Y. Chronic kidney disease and NLRP3 inflammasome: pathogenesis, development and targeted therapeutic strategies. Biochem. Biophys. Rep. 33, 101417 (2023).
Google Scholar
Shahzad, K. et al. Nlrp3-inflammasome activation in non-myeloid-derived cells aggravates diabetic nephropathy. Kidney Int. 87, 74–84 (2015).
Google Scholar
Schroder, K., Zhou, R. & Tschopp, J. The NLRP3 inflammasome: a sensor for metabolic danger? Science 327, 296–300 (2010).
Google Scholar
Tschopp, J. & Schroder, K. NLRP3 inflammasome activation: the convergence of multiple signalling pathways on ROS production? Nat. Rev. Immunol. 10, 210–215 (2010).
Google Scholar
Shahzad, K. et al. Podocyte-specific Nlrp3 inflammasome activation promotes diabetic kidney disease. Kidney Int. 102, 766–779 (2022).
Google Scholar
Nauck, M. A., Quast, D. R., Wefers, J. & Meier, J. J. GLP-1 receptor agonists in the treatment of type 2 diabetes — state-of-the-art. Mol. Metab. 46, 101102 (2021).
Google Scholar
Iqbal, J. et al. Effect of glucagon-like peptide-1 receptor agonists on body weight in adults with obesity without diabetes mellitus — a systematic review and meta-analysis of randomized control trials. Obes. Rev. 23, e13435 (2022).
Google Scholar
Yao, H. et al. Comparative effectiveness of GLP-1 receptor agonists on glycaemic control, body weight, and lipid profile for type 2 diabetes: systematic review and network meta-analysis. BMJ 384, e076410 (2024).
Google Scholar
Htike, Z. Z. et al. Efficacy and safety of glucagon-like peptide-1 receptor agonists in type 2 diabetes: a systematic review and mixed-treatment comparison analysis. Diabetes Obes. Metab. 19, 524–536 (2017).
Google Scholar
Boyle, J. G., Livingstone, R. & Petrie, J. R. Cardiovascular benefits of GLP-1 agonists in type 2 diabetes: a comparative review. Clin. Sci. 132, 1699–1709 (2018).
Google Scholar
Nauck, M. A., Meier, J. J., Cavender, M. A., Abd El Aziz, M. & Drucker, D. J. Cardiovascular actions and clinical outcomes with glucagon-like peptide-1 receptor agonists and dipeptidyl peptidase-4 inhibitors. Circulation 136, 849–870 (2017).
Google Scholar
Mann, J. F. E. et al. Potential kidney protection with liraglutide and semaglutide: Exploratory mediation analysis. Diabetes Obes. Metab. 23, 2058–2066 (2021).
Google Scholar
Mann, J. F. E. et al. Liraglutide and renal outcomes in type 2 diabetes. N. Engl. J. Med. 377, 839–848 (2017).
Google Scholar
Apperloo, E. M. et al. Tirzepatide associated with reduced albuminuria in participants with type 2 diabetes: pooled post hoc analysis from the randomized active- and placebo-controlled SURPASS-1-5 clinical trials. Diabetes Care 48, 430–436 (2025).
Google Scholar
Heerspink, H. J. L. et al. Effects of semaglutide on albuminuria and kidney function in people with overweight or obesity with or without type 2 diabetes: exploratory analysis from the step 1, 2, and 3 trials. Diabetes Care 46, 801–810 (2023).
Google Scholar
Ceriello, A. et al. Simultaneous GLP-1 and insulin administration acutely enhances their vasodilatory, antiinflammatory, and antioxidant action in type 2 diabetes. Diabetes Care 37, 1938–1943 (2014).
Google Scholar
Chaudhuri, A. et al. Exenatide exerts a potent antiinflammatory effect. J. Clin. Endocrinol. Metab. 97, 198–207 (2012).
Google Scholar
Sourris, K. C. et al. Glucagon-like peptide-1 receptor signaling modifies the extent of diabetic kidney disease through dampening the receptor for advanced glycation end products-induced inflammation. Kidney Int. 105, 132–149 (2024).
Google Scholar
Ougaard, M. E. et al. Liraglutide improves the kidney function in a murine model of chronic kidney disease. Nephron 144, 595–606 (2020).
Google Scholar
Grayson, P. C. et al. Metabolic pathways and immunometabolism in rare kidney diseases. Ann. Rheum. Dis. 77, 1226–1233 (2018).
Google Scholar
Wang, M. Y. et al. Downregulation of the kidney glucagon receptor, essential for renal function and systemic homeostasis, contributes to chronic kidney disease. Cell Metab. 36, 575–597 e577 (2024).
Google Scholar
Marso, S. P. et al. Liraglutide and cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 375, 311–322 (2016).
Google Scholar
Zavattaro, M. et al. One-year treatment with liraglutide improved renal function in patients with type 2 diabetes: a pilot prospective study. Endocrine 50, 620–626 (2015).
Google Scholar
Davies, M. J. et al. Efficacy and safety of liraglutide versus placebo as add-on to glucose-lowering therapy in patients with type 2 diabetes and moderate renal impairment (LIRA-RENAL): a randomized clinical trial. Diabetes Care 39, 222–230 (2016).
Google Scholar
Wexler, D. J. et al. Comparative effects of glucose-lowering medications on kidney outcomes in type 2 diabetes: the grade randomized clinical trial. JAMA Intern. Med. 183, 705–714 (2023).
Google Scholar
Neumiller, J. J. et al. Kidney outcomes with glucagon-like peptide-1 receptor agonists, sodium-glucose cotransporter 2 inhibitors, dipeptidyl peptidase-4 inhibitors, and sulfonylureas in type 2 diabetes and moderate cardiovascular risk. Clin. J. Am. Soc. Nephrol. 20, 206–217 (2024).
Google Scholar
Ndumele, C. E. et al. A synopsis of the evidence for the science and clinical management of cardiovascular-kidney-metabolic (CKM) syndrome: a scientific statement from the American Heart Association. Circulation 148, 1636–1664 (2023).
Google Scholar
Pfeffer, M. A. et al. Lixisenatide in patients with type 2 diabetes and acute coronary syndrome. N. Engl. J. Med. 373, 2247–2257 (2015).
Google Scholar
Muskiet, M. H. A. et al. Lixisenatide and renal outcomes in patients with type 2 diabetes and acute coronary syndrome: an exploratory analysis of the ELIXA randomised, placebo-controlled trial. Lancet Diabetes Endocrinol. 6, 859–869 (2018).
Google Scholar
Holman, R. R. et al. Effects of once-weekly exenatide on cardiovascular outcomes in type 2 diabetes. N. Engl. J. Med. 377, 1228–1239 (2017).
Google Scholar
Bethel, M. A. et al. Microvascular and cardiovascular outcomes according to renal function in patients treated with once-weekly exenatide: insights from the EXSCEL trial. Diabetes Care 43, 446–452 (2019).
Google Scholar
Muskiet, M. H. A. et al. Exenatide twice-daily does not affect renal function or albuminuria compared to titrated insulin glargine in patients with type 2 diabetes mellitus: a post-hoc analysis of a 52-week randomised trial. Diabetes Res. Clin. Pract. 153, 14–22 (2019).
Google Scholar
Gerstein, H. C. et al. Dulaglutide and cardiovascular outcomes in type 2 diabetes (REWIND): a double-blind, randomised placebo-controlled trial. Lancet 394, 121–130 (2019).
Google Scholar
Gerstein, H. C. et al. Dulaglutide and renal outcomes in type 2 diabetes: an exploratory analysis of the REWIND randomised, placebo-controlled trial. Lancet 394, 131–138 (2019).
Google Scholar
Tuttle, K. R. et al. Dulaglutide versus insulin glargine in patients with type 2 diabetes and moderate-to-severe chronic kidney disease (AWARD-7): a multicentre, open-label, randomised trial. Lancet Diabetes Endocrinol. 6, 605–617 (2018).
Google Scholar
Tuttle, K. R. et al. Clinical outcomes by albuminuria status with dulaglutide versus insulin glargine in participants with diabetes and CKD: AWARD-7 exploratory analysis. Kidney360 2, 254–262 (2021).
Google Scholar
Hernandez, A. F. et al. Albiglutide and cardiovascular outcomes in patients with type 2 diabetes and cardiovascular disease (Harmony Outcomes): a double-blind, randomised placebo-controlled trial. Lancet 392, 1519–1529 (2018).
Google Scholar
Gerstein, H. C. et al. Cardiovascular and renal outcomes with efpeglenatide in type 2 diabetes. N. Engl. J. Med. 385, 896–907 (2021).
Google Scholar
Marso, S. P. et al. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N. Engl. J. Med. 375, 1834–1844 (2016).
Google Scholar
Tuttle, K. R. et al. Effects of once-weekly semaglutide on kidney disease outcomes by KDIGO risk category in the SUSTAIN 6 trial. Kidney Int. Rep. 9, 2006–2015 (2024).
Google Scholar
Lincoff, A. M. et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N. Engl. J. Med. 389, 2221–2232 (2023).
Google Scholar
McGuire, D. K. et al. Oral semaglutide and cardiovascular outcomes in high-risk type 2 diabetes. N. Engl. J. Med. 392, 2001–2012 (2025).
Google Scholar
Heerspink, H. J. L. et al. Effects of tirzepatide versus insulin glargine on kidney outcomes in type 2 diabetes in the SURPASS-4 trial: post-hoc analysis of an open-label, randomised, phase 3 trial. Lancet Diabetes Endocrinol. 10, 774–785 (2022).
Google Scholar
Heerspink, H. J. L. et al. Effects of tirzepatide versus insulin glargine on cystatin c-based kidney function: a SURPASS-4 post hoc analysis. Diabetes Care 46, 1501–1506 (2023).
Google Scholar
Heerspink, H. J. L. et al. Kidney parameters with tirzepatide in obesity with or without type 2 diabetes. J. Am. Soc. Nephrol. 36, 2190–2200 (2025).
Google Scholar
Shaman, A. M. et al. Effect of the glucagon-like peptide-1 receptor agonists semaglutide and liraglutide on kidney outcomes in patients with type 2 diabetes: pooled analysis of SUSTAIN 6 and LEADER. Circulation 145, 575–585 (2022).
Google Scholar
Tuttle, K. R. et al. Post hoc analysis of SUSTAIN 6 and PIONEER 6 trials suggests that people with type 2 diabetes at high cardiovascular risk treated with semaglutide experience more stable kidney function compared with placebo. Kidney Int. 103, 772–781 (2023).
Google Scholar
Apperloo, E. M. et al. Effect of semaglutide on kidney function across different levels of baseline HbA1c, blood pressure, body weight and albuminuria in SUSTAIN 6 and PIONEER 6. Nephrol. Dial. Transpl. 40, 352–359 (2025).
Google Scholar
Sattar, N. et al. Cardiovascular, mortality, and kidney outcomes with GLP-1 receptor agonists in patients with type 2 diabetes: a systematic review and meta-analysis of randomised trials. Lancet Diabetes Endocrinol. 9, 653–662 (2021).
Google Scholar
Badve, S. V. et al. Effects of GLP-1 receptor agonists on kidney and cardiovascular disease outcomes: a meta-analysis of randomised controlled trials. Lancet Diabetes Endocrinol. 13, 15–28 (2025).
Google Scholar
Mendonca, L., Moura, H., Chaves, P. C., Neves, J. S. & Ferreira, J. P. The impact of glucagon-like peptide-1 receptor agonists on kidney outcomes: a meta-analysis of randomized placebo-controlled trials. Clin. J. Am. Soc. Nephrol. 20, 159–168 (2024).
Google Scholar
Lee, M. M. Y. et al. Cardiovascular and kidney outcomes and mortality with long-acting injectable and oral glucagon-like peptide 1 receptor agonists in individuals with type 2 diabetes: a systematic review and meta-analysis of randomized trials. Diabetes Care 48, 846–859 (2025).
Google Scholar
Chu, L., Bradley, R. M., Auerbach, P. & Abitbol, A. Real-world impact of adding a glucagon-like peptide-1 receptor agonist compared with basal insulin on metabolic targets in adults living with type 2 diabetes and chronic kidney disease already treated with a sodium-glucose co-transporter-2 inhibitor: the impact GLP-1 CKD study. Diabetes Obes. Metab. 26, 4674–4683 (2024).
Google Scholar
Zhang, S. et al. Healthcare utilization, mortality, and cardiovascular events following GLP1-RA initiation in chronic kidney disease. Nat. Commun. 15, 10623 (2024).
Google Scholar
Nicholas, S. B. et al. Prescription of guideline-directed medical therapies in patients with diabetes and chronic kidney disease from the CURE-CKD Registry, 2019-2020. Diabetes Obes. Metab. 25, 2970–2979 (2023).
Google Scholar
Lassen, M. C. H. et al. Adherence to glucagon-like peptide-1 receptor agonist treatment in type 2 diabetes mellitus: a nationwide registry study. Diabetes Obes. Metab. 26, 5239–5250 (2024).
Google Scholar
Jung, H. et al. Clinical characteristics, treatment patterns, and persistence in individuals with type 2 diabetes initiating a glucagon-like peptide-1 receptor agonist: a retrospective analysis of the diabetes prospective follow-up registry. Diabetes Obes. Metab. 25, 1813–1822 (2023).
Google Scholar
Sikirica, M. V. et al. Reasons for discontinuation of GLP1 receptor agonists: data from a real-world cross-sectional survey of physicians and their patients with type 2 diabetes. Diabetes Metab. Syndr. Obes. 10, 403–412 (2017).
Google Scholar
McEwan, P. et al. Cost-effectiveness of semaglutide in people with obesity and cardiovascular disease without diabetes. J. Med. Econ. 28, 268–278 (2025).
Google Scholar
Yang, C. T., Yao, W. Y., Ou, H. T. & Kuo, S. Value of GLP-1 receptor agonists versus long-acting insulins for type 2 diabetes patients with and without established cardiovascular or chronic kidney diseases: a model-based cost-effectiveness analysis using real-world data. Diabetes Res. Clin. Pract. 198, 110625 (2023).
Google Scholar
Xie, Y., Choi, T. & Al-Aly, Z. Mapping the effectiveness and risks of GLP-1 receptor agonists. Nat. Med. 31, 951–962 (2025).
Google Scholar
van Raalte, D. H. et al. Combination therapy for kidney disease in people with diabetes mellitus. Nat. Rev. Nephrol. 20, 433–446 (2024).
Google Scholar
Fu, E. L. et al. A population-based cohort defined risk of hyperkalemia after initiating SGLT-2 inhibitors, GLP1 receptor agonists or DPP-4 inhibitors to patients with chronic kidney disease and type 2 diabetes. Kidney Int. 105, 618–628 (2024).
Google Scholar
Fu, E. L. et al. SGLT-2 inhibitors, GLP-1 receptor agonists, and DPP-4 inhibitors and risk of hyperkalemia among people with type 2 diabetes in clinical practice: population based cohort study. BMJ 385, e078483 (2024).
Google Scholar
Huang, T. et al. GLP-1RA vs DPP-4i use and rates of hyperkalemia and RAS blockade discontinuation in type 2 diabetes. JAMA Intern. Med. 184, 1195–1203 (2024).
Google Scholar
Alicic, R. Z., Neumiller, J. J. & Tuttle, K. R. Combination therapy: an upcoming paradigm to improve kidney and cardiovascular outcomes in chronic kidney disease. Nephrol. Dial. Transpl. 40, i3–i17 (2025).
Google Scholar
Mann, J. F. E. et al. Effects of semaglutide with and without concomitant SGLT2 inhibitor use in participants with type 2 diabetes and chronic kidney disease in the FLOW trial. Nat. Med. 30, 2849–2856 (2024).
Google Scholar
Rossing, P. et al. Finerenone in patients across the spectrum of chronic kidney disease and type 2 diabetes by glucagon-like peptide-1 receptor agonist use. Diabetes Obes. Metab. 25, 407–416 (2023).
Google Scholar
Cherney, D. Z. I. et al. Rationale, design and baseline characteristics of REMODEL, a mechanism-of-action trial with semaglutide in people with type 2 diabetes and chronic kidney disease. Nephrol. Dial. Transpl. 40, 2182–2192 (2025).
Google Scholar
Wharton, S. et al. Daily oral GLP-1 receptor agonist orforglipron for adults with obesity. N. Engl. J. Med. 389, 877–888 (2023).
Google Scholar
Rosenstock, J. et al. Orforglipron, an oral small-molecule GLP-1 receptor agonist, in early type 2 diabetes. N. Engl. J. Med. 393, 1065–1076 (2025).
Google Scholar
Gasiorek, A. et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of the first-in-class GLP-1 and amylin receptor agonist, amycretin: a first-in-human, phase 1, double-blind, randomised, placebo-controlled trial. Lancet 406, 135–148 (2025).
Google Scholar
Dahl, K. et al. Amycretin, a novel, unimolecular GLP-1 and amylin receptor agonist administered subcutaneously: results from a phase 1b/2a randomised controlled study. Lancet 406, 149–162 (2025).
Google Scholar
Selvarajah, V. et al. A randomized phase 2b trial examined the effects of the glucagon-like peptide-1 and glucagon receptor agonist cotadutide on kidney outcomes in patients with diabetic kidney disease. Kidney Int. 106, 1170–1180 (2024).
Google Scholar
Tuttle, K. R. et al. Clinical characteristics of and risk factors for chronic kidney disease among adults and children: an analysis of the CURE-CKD registry. JAMA Netw. Open. 2, e1918169 (2019).
Google Scholar
Norris, K. C. et al. Rationale and design of a multicenter chronic kidney disease (CKD) and at-risk for CKD electronic health records-based registry: CURE-CKD. BMC Nephrol. 20, 416 (2019).
Google Scholar
Nee, R., Yuan, C. M., Narva, A. S., Yan, G. & Norris, K. C. Overcoming barriers to implementing new guideline-directed therapies for chronic kidney disease. Nephrol. Dial. Transpl. 38, 532–541 (2023).
Google Scholar
Exenatide injection. Prescribing information (Amneal Pharmaceuticals, 2025).
Liraglutide (Victoza) injection. Prescribing information (Novo Nordisk, 2025).
Liraglutide (Saxenda) injection. Prescribing information (Novo Nordisk, 2025).
Exenatide extended-release (Bydureon BCise) injectable suspension. Prescribing information (AstraZeneca Pharmaceuticals, 2025).
Dulaglutide (Trulicity) injection. Prescribing information (Eli Lilly, 2024).
Semaglutide (Ozempic) injection. Prescribing information (Novo Nordisk, 2025).
Semaglutide (Wegovy) injection. Prescribing information (Novo Nordisk, 2025).
Semaglutide (Rybelsus) tablets. Prescribing information (Novo Nordisk, 2025).
Tirzepatide (Mounjaro) injection. Prescribing information (Eli Lilly, 2025).
Tirzepatide (Zepbound) injection. Prescribing information (Eli Lilly, 2025).
Del Prato, S. et al. Tirzepatide versus insulin glargine in type 2 diabetes and increased cardiovascular risk (SURPASS-4): a randomised, open-label, parallel-group, multicentre, phase 3 trial. Lancet 398, 1811–1824 (2021).
Google Scholar
Holst, J. J. et al. Proglucagon processing in porcine and human pancreas. J. Biol. Chem. 269, 18827–18833 (1994).
Google Scholar
Drucker, D. J. & Asa, S. Glucagon gene expression in vertebrate brain. J. Biol. Chem. 263, 13475–13478 (1988).
Google Scholar
Portela-Gomes, G. M. & Stridsberg, M. Chromogranin A in the human gastrointestinal tract: an immunocytochemical study with region-specific antibodies. J. Histochem. Cytochem. 50, 1487–1492 (2002).
Google Scholar
Mortensen, K., Christensen, L. L., Holst, J. J. & Orskov, C. GLP-1 and GIP are colocalized in a subset of endocrine cells in the small intestine. Regul. Pept. 114, 189–196 (2003).
Google Scholar
Ugleholdt, R. et al. Prohormone convertase 1/3 is essential for processing of the glucose-dependent insulinotropic polypeptide precursor. J. Biol. Chem. 281, 11050–11057 (2006).
Google Scholar
Baggio, L. L. & Drucker, D. J. Biology of incretins: GLP-1 and GIP. Gastroenterology 132, 2131–2157 (2007).
Google Scholar
Cheroutre, H., Lambolez, F. & Mucida, D. The light and dark sides of intestinal intraepithelial lymphocytes. Nat. Rev. Immunol. 11, 445–456 (2011).
Google Scholar
Martchenko, S. E. et al. Circadian GLP-1 Secretion in mice is dependent on the intestinal microbiome for maintenance of diurnal metabolic homeostasis. Diabetes 69, 2589–2602 (2020).
Google Scholar
Yoon, H. S. et al. Akkermansia muciniphila secretes a glucagon-like peptide-1-inducing protein that improves glucose homeostasis and ameliorates metabolic disease in mice. Nat. Microbiol. 6, 563–573 (2021).
Google Scholar
Gribble, F. M. & Reimann, F. Enteroendocrine cells: chemosensors in the intestinal epithelium. Annu. Rev. Physiol. 78, 277–299 (2016).
Google Scholar
Vilsboll, T. et al. Incretin secretion in relation to meal size and body weight in healthy subjects and people with type 1 and type 2 diabetes mellitus. J. Clin. Endocrinol. Metab. 88, 2706–2713 (2003).
Google Scholar
Furness, J. B., Rivera, L. R., Cho, H. J., Bravo, D. M. & Callaghan, B. The gut as a sensory organ. Nat. Rev. Gastroenterol. Hepatol. 10, 729–740 (2013).
Google Scholar
Gribble, F. M. & Reimann, F. Function and mechanisms of enteroendocrine cells and gut hormones in metabolism. Nat. Rev. Endocrinol. 15, 226–237 (2019).
Google Scholar
Worthington, J. J., Reimann, F. & Gribble, F. M. Enteroendocrine cells-sensory sentinels of the intestinal environment and orchestrators of mucosal immunity. Mucosal Immunol. 11, 3–20 (2018).
Google Scholar
Berthier, C. C. et al. Enhanced expression of Janus kinase-signal transducer and activator of transcription pathway members in human diabetic nephropathy. Diabetes 58, 469–477 (2009).
Google Scholar
Peng, Y. et al. AGE-RAGE signal generates a specific NF-κB RelA “barcode” that directs collagen I expression. Sci. Rep. 6, 18822 (2016).
Google Scholar
Qi, C., Mao, X., Zhang, Z. & Wu, H. Classification and differential diagnosis of diabetic nephropathy. J. Diabetes Res. 2017, 8637138 (2017).
Google Scholar
Fujita, H. et al. The protective roles of GLP-1R signaling in diabetic nephropathy: possible mechanism and therapeutic potential. Kidney Int. 85, 579–589 (2014).
Google Scholar
link










More Stories
Russia turns to high altitude balloons after Starlink restrictions disrupt battlefield communications across Russian-controlled territories
What CES 2026 didn’t show: The quiet crisis in wireless capacity nobody is talking about
Recent AWS issues blamed on AI tools – at least two incidents affected some Amazon services